BASKING RIDGE, N.J., Jan. 25, 2021 (GLOBE NEWSWIRE) — Caladrius Biosciences, Inc. (Nasdaq: CLBS) (“Caladrius” or the “Company”), a clinical-stage biopharmaceutical company dedicated to the development of cellular therapies designed to reverse disease, today announced that it has closed on its previously announced sale of an aggregate of 12,500,000 shares of […]
Other News
Amarin Expands Cardiovascular Risk Reduction Patent Infringement Lawsuit to Include Health Care Insurance Provider
DUBLIN, Ireland and BRIDGEWATER, N.J., Jan. 25, 2021 (GLOBE NEWSWIRE) — Amarin Corporation plc (NASDAQ:AMRN), announced today an expansion of the scope of its VASCEPA® (icosapent ethyl) cardiovascular (CV) risk reduction patent infringement lawsuit against Hikma Pharmaceuticals PLC to include a health care insurance provider in the United States, Health Net, […]
12-Month Data from Surmodics’ TRANSCEND Trial Presented at LINC 2021 Event
SurVeil™ Drug Coated Balloon (DCB) demonstrates non-inferior safety and efficacy, while using a substantially lower drug dose, vs. the IN.PACT® Admiral® DCB for treatment of femoropopliteal lesions. EDEN PRAIRIE, Minn.–(BUSINESS WIRE)–Surmodics, Inc. (NASDAQ:SRDX), a leading provider of medical device and in vitro diagnostic technologies to the health care industry, today announced that […]
Kardium Announces $115M in New Financing for Innovative Atrial Fibrillation Treatment
Fidelity Management & Research Company LLC and T. Rowe Price funds lead private financing round VANCOUVER, British Columbia–(BUSINESS WIRE)–Kardium Inc., developer of the Globe® Mapping and Ablation System for the treatment of atrial fibrillation, has raised US $115 million in a new financing round. The round is led by Fidelity Management & Research […]
Alleviant Medical Receives Breakthrough Device Designation From FDA for Transcatheter Technology
Innovative technology offers an implant-free approach for individuals with chronic heart failure AUSTIN, Texas–(BUSINESS WIRE)–Alleviant Medical Inc., a privately-held medical device company, today announced that the US Food and Drug Administration (FDA) has granted the company a Breakthrough Device designation for its transcatheter technology. The technology offers a no-implant interatrial […]
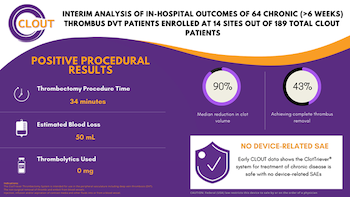
Inari Medical Announces Presentation of Positive Chronic Clot Subanalysis Results from Real World CLOUT Registry at LINC 2021
IRVINE, Calif., Jan. 25, 2021 (GLOBE NEWSWIRE) — Inari Medical, Inc. (NASDAQ: NARI) (“Inari”) a commercial-stage medical device company focused on developing products to treat and transform the lives of patients suffering from venous diseases, today announced strongly positive interim results of the first 64 chronic deep vein thrombosis (“DVT”) […]
PEDRA™ Technology Receives FDA Breakthrough Device Designation for its PEDRA™ Xauron™ Real-Time Tissue Perfusion System
Novel perfusion monitor achieves FDA Breakthrough Device Designation for real-time, periprocedural monitoring of tissue perfusion in patients with critical limb threatening ischemia SINGAPORE, Jan 25, 2021 /PRNewswire/ — PEDRA™ Technology, a privately-held company, announced today that the U.S Food and Drug Administration (FDA) has granted the company a Breakthrough Device Designation for the […]
Individual patient data meta-analysis of the effects of the CARILLON® mitral contour system
ESC Heart Failure Giallauria et al. December 2020 An individual patient data meta‐analysis was conducted from 3 prospective studies that enrolled 209 heart failure patients with reduced ejection fraction patients and functional mitral regurgitation (FMR). Patients implanted with the Carillon Mitral Contour System or control were assessed for 12-month outcomes […]
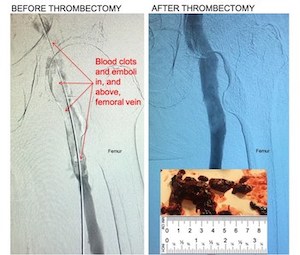
Kishor Vora, M.D. Owensboro Heart & Vascular, Removes Large Blood Clots in 1st Global Use of New Device
11F mechanical thrombectomy system removes large blood clots in deep vein thrombosis (DVT) HALLANDALE BEACH, Flo., Jan. 22, 2021 /PRNewswire/ — Control Medical Technology announced the FDA cleared Control 11F Mechanical Thrombectomy system was used to remove large blood clots from patients with deep vein thrombosis (DVT). “Control removed large blood […]
Boston Scientific Announces Agreement To Acquire Preventice Solutions, Inc.
Acquisition of external cardiac monitoring technologies and services provider to expand rhythm management diagnostics portfolio and capabilities MARLBOROUGH, Mass., Jan. 21, 2021 /PRNewswire/ — Boston Scientific (NYSE: BSX) today announced that it has entered into a definitive agreement to acquire Preventice Solutions, Inc., a privately-held company which offers a full portfolio of mobile cardiac […]