TRANSLATE-HF study establishes the potential for broad use of the only SGLT2 inhibitor indicated in patients with heart failure with reduced ejection fraction with and without diabetes WILMINGTON, Del.–(BUSINESS WIRE)–New data from a large, contemporary US hospitalized heart failure (HF) registry confirms that four out of five (81.1%) patients with […]
Other News
Vida Health Launches Virtual Congestive Heart Failure Management Program
The new program will launch for members from Vida’s national payer customers in December of this year with a full rollout in January of 2021 SAN FRANCISCO–(BUSINESS WIRE)–Expanding on its commitment to improve health outcomes for millions of lives, Vida Health announced a new app-based, coach-centered virtual congestive heart failure (CHF) management […]
PhaseBio Reports Recent Business Highlights and Third-Quarter 2020 Financial Results
Expanded pivotal Phase 3 REVERSE-IT trial of product candidate bentracimab (PB2452) for reversal of antiplatelet effects of ticagrelor into Canada and dosed first patients outside the United States Resuming enrollment in ongoing Phase 2b trial of pemziviptadil (PB1046) in pulmonary arterial hypertension MALVERN, Pa. & SAN DIEGO–(BUSINESS WIRE)–PhaseBio Pharmaceuticals, Inc. (Nasdaq: […]
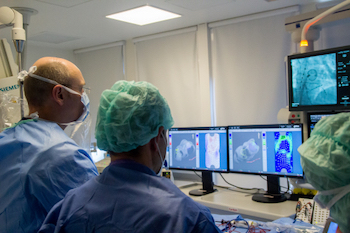
RapidAI Celebrates International Adoption of AI-Powered Stroke Care with Nearly 300% Growth
SAN MATEO, Calif.–(BUSINESS WIRE)–RapidAI, the leader in advanced imaging for stroke, today announced unprecedented growth in the adoption and use of the Rapid platform around the world. Now approved in over 60 countries and utilized in over 1,600 hospitals, the company has experienced a year-over-year increase in just International Rapid […]
Kardium Performs First Cases with the Globe® Positioning System
New 3D mapping and navigation feature allows for high-definition mapping of the full atrium VANCOUVER, British Columbia–(BUSINESS WIRE)–Kardium is pleased to announce the first clinical use of the Globe Positioning System (GPS™) – the new 3D mapping and navigation feature of its flagship product, the Globe® Mapping and Ablation System. […]
AngioDynamics Announces Presentation of Positive Safety, Efficacy Results from RAPID Outcomes Database
Represents Largest Catheter Based Thromboaspiration Study Completed to Date LATHAM, N.Y.–(BUSINESS WIRE)–AngioDynamics, Inc. (NASDAQ: ANGO), a leading provider of innovative, minimally invasive medical devices for vascular access, peripheral vascular disease, and oncology, announced the safety and efficacy results from the Registry of AngioVac* System Procedures In Detail (RAPID) database. Results […]
UNC Medical Center First in the Carolinas to Treat Heart Failure Patients Using Breakthrough CCM Therapy
Innovative minimally-invasive therapy is proven to help people with heart failure feel better CHAPEL HILL, N.C.–(BUSINESS WIRE)–UNC Medical Center today announced it is among the first hospitals in the United States and the first hospital in the Carolinas to use CCM® therapy, delivered by the Optimizer® system, to treat patients suffering from heart failure. […]
CB Scientific, Inc. (CBSC) Announces Definitive Agreement to Acquire Commercial Ambulatory ECG Device Manufacturer Datrix, LLC
Acquisition will expand Ambulatory Remote Cardiac ECG Monitoring product technology offering and outreach into US and international markets ESCONDIDO, Calif., Nov. 12, 2020 (GLOBE NEWSWIRE) — via InvestorWire – CB Scientific. Inc. (OTC:CBSC) (“CBSC” or the “Company”), a provider of innovative products and services for the ambulatory noninvasive cardiac monitoring […]
Esperion Announces Pricing of Offering of $250.0 Million of Convertible Senior Subordinated Notes
ANN ARBOR, Mich., Nov. 12, 2020 (GLOBE NEWSWIRE) — Esperion (NASDAQ: ESPR) today announced the pricing of $250.0 million aggregate principal amount of 4.00% Convertible Senior Subordinated Notes due 2025 (the “notes”) in a private offering (the “offering”) to qualified institutional buyers pursuant to Rule 144A under the Securities Act […]
Resverlogix Announces Publication in High-impact, Peer-reviewed Journal, Clinical Epigenetics
Publication Explores the Beneficial Effects of Apabetalone on the Immune Cells of Diabetic Patients Event Reminder: Multiple Presentations on Apabetalone to be made during the American Heart Association Scientific Sessions – November 13-17 CALGARY, Alberta, Nov. 12, 2020 (GLOBE NEWSWIRE) — Resverlogix Corp. (“Resverlogix” or the “Company”) (TSX:RVX) announced today the recent publication of an article titled: “BET protein inhibitor […]