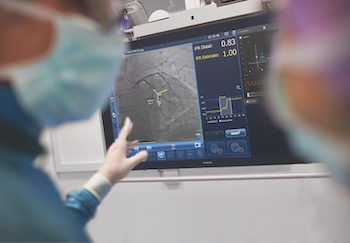
Randomized controlled trial with up to 3,000 participants at 75 sites to compare outcomes between physiological guidance by iFR data co-registered on the angiogram (interventional X-ray image) versus the standard of care use of an angiogram alone Percutaneous coronary intervention (PCI) is one of the main treatment options to open […]
Other News
PhaseBio Announces European Regulatory Update for PB2452
PB2452 granted PRIME Designation by the European Medicines Agency PhaseBio receives written scientific advice confirming PB2452 clinical development plan In Phase 1 and Phase 2a clinical trials, PB2452 provided immediate and sustained reversal of the antiplatelet effects of ticagrelor MALVERN, Pa. and SAN DIEGO, Feb. 11, 2020 (GLOBE NEWSWIRE) — PhaseBio […]
VADovations, Inc. Secures Investment and Development Agreement for a Percutaneous Endovascular Delivery System for its Next-Generation Cardiac Assist Device for Long-Term Use
OKLAHOMA CITY–(BUSINESS WIRE)–VADOVATIONS, Inc., a company developing miniaturized cardiac assist pumps offering best-in-class blood handling properties, high performance and low cost, today announces an agreement with a strategic partner to both invest in the Company and develop a percutaneous endovascular delivery system for VADovations’ novel cardiac assist pump for long-term use […]
CorWave Appoints Jim Schuermann as an Independent Director
Medtech industry veteran of two IPOs on the Nasdaq Stock Market (HeartWare, Avedro) CLICHY, France–(BUSINESS WIRE)–CorWave, a medtech company focused on the development of an innovative Left Ventricular Assist Device (LVAD) to treat patients suffering from severe heart failure, announced today the election of Jim Schuermann to its board of […]
JenaValve Technology Closes $50 Million Financing
IRVINE, Calif.–(BUSINESS WIRE)–JenaValve Technology, Inc., developer and manufacturer of the JenaValve Pericardial Transcatheter Aortic Valve Replacement (TAVR) System for the treatment of aortic valve disease, announces that it has raised $50 million in an equity financing led by Bain Capital Life Sciences. Additional participants in the financing included existing investors Andera […]
AngioDynamics Appoints Stephen Trowbridge Executive Vice President and Chief Financial Officer
AngioDynamics, Inc. (NASDAQ: ANGO), a leading provider of innovative, minimally invasive medical devices for vascular access, peripheral vascular disease, and oncology, today announced that it has appointed Stephen Trowbridge to the role of Executive Vice President and Chief Financial Officer, effective February 5, 2020. Mr. Trowbridge has served as the […]
Hemostemix Announces the Appointment of Dr. Ronnie Hershman to the Board of Directors and Provides a Corporate Update
CALGARY, Alberta, Feb. 10, 2020 (GLOBE NEWSWIRE) — Hemostemix Inc. (“Hemostemix” or the “Company”) (TSXV: HEM; OTC: HMTXF) is pleased to announce the appointment of Dr. Ronnie Hershman, M.D., F.C.C.S., to its Board of Directors. Dr. Hershman is a successful, practicing cardiologist with over three decades of experience. Dr. Hershman […]
LeMaitre Vascular Announces Q4 2019 Financial Results
February 6, 2020 at 4:05 PM EST PDF Version BURLINGTON, Mass., Feb. 06, 2020 (GLOBE NEWSWIRE) — LeMaitre Vascular, Inc. (Nasdaq:LMAT), a provider of vascular devices, implants and services, today reported Q4 2019 results, provided guidance, and announced a $0.095/share dividend. Q4 2019 Results Record sales of $30.2mm, +6% (flat organic) vs. Q4 2018 Operating income […]
CorMatrix® Cardiovascular, Inc. receives FDA approval to expand and enroll 20 additional patients in the adult arm of its early feasibility IDE study of the Cor® TRICUSPID ECM® valve for pediatric and adult patients
ATLANTA, Feb. 7, 2020 /PRNewswire/ — CorMatrix®Cardiovascular, Inc. www.cormatrix.com, a leading developer of regenerative cardiovascular medical devices, today announced receiving FDA approval to expand and enroll 20 additional patients in the adult arm, including up to 4 additional cardiac surgery research and investigative centers, for its early feasibility IDE study of the Cor® TRICUSPID ECM® valve* for […]
Amarin Honored by BioNJ with Innovator Award for VASCEPA® Development and Regulatory Approval
DUBLIN, Ireland and BRIDGEWATER, N.J., Feb. 06, 2020 (GLOBE NEWSWIRE) — Amarin Corporation plc (NASDAQ:AMRN), announced today that BioNJ awarded Amarin with an Innovator Award in recognition of the approval of a new indication for VASCEPA® (icosapent ethyl) by the U.S. Food and Drug Administration (FDA) in December 2019. This […]