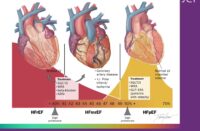
At 1 year, outcomes similar to overall cohort of patients
IRVINE, Calif., May 18, 2021 /PRNewswire/ — Edwards Lifesciences (NYSE: EW) today announced the results of a real-world study comparing outcomes for patients with bicuspid aortic stenosis (AS) who were treated with SAPIEN 3 and SAPIEN 3 Ultra Transcatheter Aortic Valve Replacement (TAVR) and at low risk of death from surgery. The data show excellent outcomes at one year, with low rates of death and stroke, and no significant differences in the primary outcomes compared with the overall cohort, or those with tricuspid aortic valve disease.
The propensity-matched analysis comparing real-world data of bicuspid and tricuspid AS patients collected from the Society of Thoracic Surgeons and American College of Cardiology (STS/ACC) Transcatheter Valve Therapy (TVT) Registry was presented today during the EuroPCR 2021 late-breaking clinical trials hotline session by Raj Makkar, M.D., Director, Interventional Cardiology, Associate Director, Smidt Heart Institute at Cedars Sinai Medical Center.
“I am encouraged to see these data reflect the favorable outcomes we’ve witnessed to date in bicuspid patients treated with SAPIEN 3 TAVR, including very low rates of death and stroke, significant and similar improvements in functional status and quality of life, and high procedural success rates,” Makkar said. “With bicuspid aortic valve disease as another anatomical consideration in the treatment of TAVR patients, these SAPIEN 3 data are encouraging and crucial for guiding treatment, particularly for younger patients.”
In a propensity-matched, real-world registry analysis of more than 6,000 patients treated with SAPIEN 3 TAVR at low surgical risk, the all-cause mortality rate for bicuspid valves was low and similar at 1 year when compared to those with tricuspid valve disease (4.6% versus 6.6%) as were stroke rates at 1 year (2.0% versus 2.1%). In addition to the excellent survival and stroke rates, outcomes data from the TVT Registry also demonstrated a low rate of significant paravalvular leak, as well as significant and similar improvements in functional status and quality of life measures.
“These SAPIEN 3 data provide further evidence and confidence for physicians in treating their patients with bicuspid valves who prefer TAVR as an option to open-heart surgery,” said Larry L. Wood, Edwards’ corporate vice president, transcatheter aortic valve replacement. “This supports recent American College of Cardiology/American Heart Association guidelines that recommend the importance of shared decision-making with patients based on the robust clinical evidence that show both TAVR and surgical treatment options should be considered for all patients 65 years and older.”
The SAPIEN 3 valve builds on Edwards’ decades of experience in the development of tissue heart valves, and the proven benefits of the Edwards SAPIEN valves. The Edwards SAPIEN family of valves are the most studied transcatheter heart valves in history and have been used in the treatment of thousands of patients globally.
Makkar is a consultant to Edwards Lifesciences.
About Edwards Lifesciences
Edwards Lifesciences is the global leader of patient-focused innovations for structural heart disease and critical care monitoring. We are driven by a passion for patients, dedicated to improving and enhancing lives through partnerships with clinicians and stakeholders across the global healthcare landscape. For more information, visit Edwards.com and follow us on Facebook, Instagram, LinkedIn, Twitter and YouTube.
This news release includes forward-looking statements within the meaning of Section 27A of the Securities Act of 1933 and Section 21E of the Securities Exchange Act of 1934. These forward-looking statements include, but are not limited to, statements by Dr. Makkar and Mr. Wood and statements regarding expected product and program benefits and clinical outcomes. Forward-looking statements are based on estimates and assumptions made by management of the company and are believed to be reasonable, though they are inherently uncertain and difficult to predict. Our forward-looking statements speak only as of the date on which they are made and we do not undertake any obligation to update any forward-looking statement to reflect events or circumstances after the date of the statement. Statements of past performance, efforts, or results about which inferences or assumptions may be made can also be forward-looking statements and are not indicative of future performance or results; these statements can be identified by the use of words such as “continued,” “transform,” “develop,” “preliminary,” “initial,” “diligence,” “industry-leading,” “compliant,” “indications,” or “early feedback” or other forms of these words or similar words or expressions or the negative thereof. Investors are cautioned not to unduly rely on such forward-looking statements.
Forward-looking statements involve risks and uncertainties that could cause results to differ materially from those expressed or implied by the forward-looking statements based on a number of factors including but not limited to unexpected outcomes after more expanded clinical experience, unexpected changes or delays related to product supply, potentials for unexpected regulatory or quality developments, competitive dynamics, or unexpected delays or changes in patient access, litigation or clinician acceptance. Other factors are detailed in the company’s filings with the Securities and Exchange Commission including its Annual Report on Form 10-K for the year ended December 31, 2020 and the Quarterly Report on Form 10-Q for the quarter ended March 31, 2021. These filings, along with important safety information about our products, may be found at edwards.com.
Edwards, Edwards Lifesciences, the stylized E logo, Edwards SAPIEN, Edwards SAPIEN 3, SAPIEN, SAPIEN 3, and SAPIEN 3 Ultra are trademarks of Edwards Lifesciences Corporation. All other trademarks are the property of their respective owners. This statement is made on behalf of Edwards Lifesciences Corporation and its subsidiaries.
SOURCE Edwards Lifesciences Corporation