SAN CLEMENTE, Calif.–(BUSINESS WIRE)–Reflow Medical, Inc., a California-based medical device company, has completed enrollment in the DEEPER LIMUS clinical trial (NCT04162418) to evaluate the Temporary Spur Stent System, a patented device designed to treat long, diffuse and severely calcified infrapopliteal disease. The system allows for uniform expansion of the stent […]
Tag: ReFlow Medical
Enrollment Completed in Reflow Medical’s DEEPER OUS Clinical Trial
SAN CLEMENTE, Calif.–(BUSINESS WIRE)–Reflow Medical, Inc. announces that it has completed patient enrollment in the DEEPER OUS clinical trial (NCT03807531) for the company’s Temporary Spur Stent System, a novel device that features a retrievable stent designed for complex infrapopliteal disease. 106 patients have now been enrolled in the prospective, nonrandomized […]
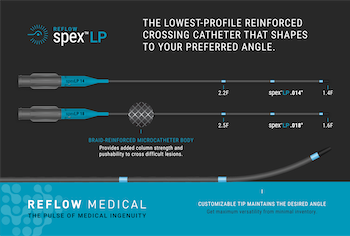
Reflow Medical Introduces the Spex™ LP, the Lowest Profile Shapeable Reinforced Support Catheter
SAN CLEMENTE, Calif.–(BUSINESS WIRE)–Reflow Medical, Inc., a California-based medical device company, introduces the Reflow™ Spex™ LP (Low Profile) 0.014 and 0.018-inch reinforced support catheters. The new Spex LP is engineered to provide the lowest profile tip for accessing and crossing the tightest and most complex lesions with a supportive system. It also […]
Reflow Medical Enrolls First Patients in DEEPER OUS Spur Stent Trial
San Clemente, Calif., July 30th, 2019–(BUSINESS WIRE) —Reflow Medical, Inc. announces that the first patients have been enrolled in its DEEPER OUS clinical trial using the Temporary Spur Stent System. DEEPER OUS is a 100-patient prospective, non-randomized, multicenter trial designed to assess the safety and efficacy of the Temporary Spur Stent […]
Reflow Medical announces enrollment of its first patients in the Wing-IT IDE clinical trial
San Clemente, Calif., March 12, 2018 /PRNewswire/ — Reflow Medical Inc., a developer of innovative medical devices to combat cardiovascular disease, announced that the first patients have been enrolled in a prospective, multi-center, non-randomized study intended to evaluate the ability of the Reflow Wingman Catheters to cross chronic total occlusions […]
Reflow Medical Announces 510(k) Clearance to Market Wingman Crossing Catheter for Coronary Indication
SAN CLEMENTE, Calif.–(BUSINESS WIRE)–Reflow Medical, Inc., a developer and innovator of crossing and support catheters for use in the treatment of occlusive cardiovascular disease, announced that the company has received 510(k) clearance from the U.S. Food and Drug Administration (FDA) to market its Wingman14C Crossing Catheter for use in conjunction […]