VANCOUVER, British Columbia, Feb. 05, 2025 (GLOBE NEWSWIRE) — Algernon Pharmaceuticals Inc. (the “Company” or “AGN Pharma”) (CSE: AGN) (FRANKFURT: AGW0) (OTCQB: AGNPF), a Canadian clinical stage pharmaceutical development company, is pleased to announce that its wholly owned subsidiary, Algernon NeuroScience (AGN Neuro), has appointed Validcare as the contract research organization (“CRO”) for the Company’s upcoming randomized, double-blind, placebo-controlled Phase 2a DMT study of 40 stroke patients in Europe. The study is expected to begin enrolling patients in Q3 of 2025. Validcare is a leading U.S. based full service CRO with experience across a wide range of therapeutic indications.
Tag: stroke
RapidPulse Receives FDA Approval to Begin IDE Study in Ischemic Stroke Using Innovative Cyclic Aspiration System – Recently Completed Clinical Trial Shows Significantly Higher First Pass and Frontline Success Rates
MIAMI, Dec. 11, 2024 (GLOBE NEWSWIRE) — RapidPulse, Inc., a Delaware corporation headquartered in Miami, Florida focused on improving the treatment success rate for acute ischemic stroke (AIS) announced that the United States Food and Drug Administration (FDA) agreed that RapidPulse can begin enrollment in an Investigational Device Exemption (IDE) study of its novel and precise cyclic aspiration system. This will allow U.S. and European patient enrollment in 2025 to commence in a study designed to demonstrate the RapidPulse patented method of precisely pulsed aspiration is safe and effective in the treatment of AIS. RapidPulse’s technology is a spin-out of Syntheon 2.0, LLC, an innovative medical device incubator with a long track record of successful exits.
Genomadix Inc. Enters into Collaboration with Mayo Clinic on Precision Medicine for Telestroke
OTTAWA, ON, Nov. 15, 2024 /PRNewswire/ – Genomadix (genomadix.com), a global leader in point of need precision medicine diagnostics, announced today that it has entered into a know-how license agreement with Mayo Clinic to collaborate on the delivery of enhanced telestroke services in the…
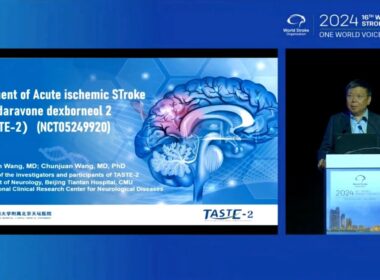
TASTE-2 World Stroke Congress | Sanbexin ® combined with thrombectomy significantly reduces stroke disability
NANJING, China, Oct. 25, 2024 /PRNewswire/ — On October 24, 2024, TASTE-2, a Chinese innovative stroke drug study led by Professor Wang Yongjun from Beijing Tiantan Hospital, was published in an oral report at the 16th World Stroke Conference (WSC): brain cell protection combined with…
Remedy Pharmaceuticals Announces FDA Orphan Drug Designation Granted for the Treatment of Large Territory Acute Ischemic Stroke with CIRARA
NEW YORK, Oct. 22, 2024 /PRNewswire/ — Remedy Pharmaceuticals, a pioneer in stroke drug development, today announced that the U.S. Food and Drug Administration (FDA) Office of Orphan Products Division (OOPD) has granted Orphan Drug Designation for CIRARA for the treatment of “large…
KFSHRC Launches Mobile Stroke Unit to Accelerate Patient Treatment
A First in the Middle East and North Africa
KFSHRC Launches Mobile Stroke Unit to Accelerate Patient Treatment
Stryker completes acquisition of NICO Corporation, expanding minimally invasive solutions for brain tumor removal and stroke care
PORTAGE, Mich., USA, Sept. 20, 2024 /PRNewswire/ — Stryker (NYSE: SYK), a global leader in medical technologies, announced today that it has completed the acquisition of NICO Corporation, a privately held company providing a systematic approach to minimally invasive surgery for tumor and…
Breakthrough Therapy designation for Sanbexin sublingual tablets granted by the United States Food and Drug Administration
NANJING, China, Sept. 5, 2024 /PRNewswire/ — On September 2, 2024, Simcere Pharmaceuticals Group Ltd. (2096.HK) announced that Sanbexin Sublingual Tablets (Edaravone and Dexborneol sublingual tablets), an innovative drug for stroke, has been granted the Breakthrough Therapy designation…
Bioxodes reports positive DMC meeting for BIOX-101 Phase 2a trial in intracerebral hemorrhagic stroke
Gosselies (Belgium), August 21, 2024 – Bioxodes SA, a clinical stage biopharmaceutical company developing novel therapies to prevent and treat thrombotic and inflammatory diseases, announces today that the DMC reviewed the data from the first 8 patients in a planned review of the ongoing BIRCH Phase 2a trial of BIOX-101 to prevent secondary damage after intracerebral hemorrhagic stroke (ICH).
MemorialCare’s Long Beach Medical Center and Saddleback Medical Center Nationally Recognized by the American Heart Association with Get with the Guidelines Gold Plus Awards
FOUNTAIN VALLEY, Calif., Aug. 20, 2024 /PRNewswire/ — MemorialCare’s Long Beach Medical Center and Saddleback Medical Center received the American Heart Association’s Get With The Guidelines® – Stroke Gold Plus quality achievement awards. Long Beach Medical Center also received the Get…