ST. LOUIS, April 06, 2026 (GLOBE NEWSWIRE) — Stereotaxis (NYSE: STXS), a pioneer and global leader in surgical robotics for minimally invasive endovascular intervention, today announced that it has received U.S. Food and Drug Administration 510(k) clearance for the Synchrony™ system. Synchrony is designed to digitize and modernize the interventional cath lab. Synchrony’s slim and stunning 55” 4K ultra-high-definition display consolidates the viewing and control of all disparate systems in the lab, offering an enhanced procedure experience with custom layouts, streamlined workflows, an intuitive user interface, and a decluttered environment. Synchrony digitizes the video streams with full fidelity and ultra-low latency, offering crystal-clear visualization. Its architecture allows obsolescence protection for labs as new technologies are introduced in the future. Synchrony is made available with SynX™, a cloud-based HIPAA and GDPR-compliant app that allows for secure remote connectivity, collaboration, recording, and monitoring of the cath lab. “We have long recognized that seamless remote connectivity and collaboration for our EP labs could be very beneficial,” said Dr. Mauricio Arruda, Professor of Medicine at Case Western Reserve University School of Medicine and Director of Electrophysiology at University Hospitals Harrington Heart & Vascular Institute. “We are particularly interested in being early pioneers of Synchrony and SynX, and look forward to demonstrating that intraoperative collaboration enhances our ability to provide the best patient care and train the next generation of physicians.” “We are excited by this technology and the opportunity to be among the first to modernize our catheter labs with Synchrony and SynX when the Richard M. Schulze Surgical and Critical Care Center at Abbott Northwestern Hospital opens in late August,” said Daniel Melby, MD, Medical Director of Electrophysiology Labs at Allina Health Minneapolis Heart Institute. “Our cardiology labs have become increasingly busy and complex environments, accentuating the value of technology that enhances procedure workflow, improves lab efficiency and encourages collaboration.” “In my role managing the technology across our electrophysiology labs and advising labs across the country, I have significant experience with various interventional lab display offerings,” said Matthew Dare, CEPS, Research and Technology Coordinator, Texas Cardiac Arrhythmia Institute at St. David’s Medical Center in Austin, Texas. “The underlying hardware and software architecture of Synchrony and SynX is far beyond what anyone else has developed. It promises a better intraoperative experience for physicians and nurses, improved equipment reliability and maintenance, and attractive tools for managing a cardiovascular program with remote monitoring, collaboration and recordings.” Stereotaxis is reiterating its guidance of over $3 Million in revenue this year from Synchrony systems placed independent of robotic systems. Synchrony and SynX have been engineered to be foundational platforms for future innovations. The advanced architecture enables future applications and the leveraging of artificial intelligence for enhanced clinical insights, automation, and safety. “Synchrony and SynX are central to our digital surgery efforts to modernize the interventional lab with enhanced workflow, remote connectivity, and smart AI capabilities,” said David Fischel Stereotaxis Chairman and CEO. “The technology improves the robotic cockpit, and will be critical in supporting robotic efforts for remote long-distance procedures and automated catheter navigation. The opportunity is much broader than robotic labs as we believe all cath labs stand to benefit from improved workflow, connectivity, and collaboration.” About StereotaxisStereotaxis (NYSE: STXS) is a pioneer and global leader in innovative surgical robotics for minimally invasive endovascular intervention. Its mission is the discovery, development and delivery of robotic systems, instruments, and information solutions for the interventional laboratory. These innovations help physicians provide unsurpassed patient care with robotic precision and safety, expand access to minimally invasive therapy, and enhance the productivity, connectivity, and intelligence in the operating room. Stereotaxis technology has been used to treat over 150,000 patients across the United States, Europe, Asia, and elsewhere. For more information, please visit www.stereotaxis.com. This press release includes statements that may constitute “forward-looking” statements within the meaning of Section 27A of the Securities Act of 1933 and Section 21E of the Securities Act of 1934, including statements regarding the completion of the Company’s offering and the anticipated use of proceeds therefrom, usually containing the words “believe”, “estimate”, “project”, “expect” or similar expressions. Forward-looking statements inherently involve risks and uncertainties that could cause actual results to differ materially. Factors that would cause or contribute to such differences include, but are not limited to, the Company’s ability to manage expenses at sustainable levels, acceptance of the Company’s products in the marketplace, the effect of global economic conditions on the ability and willingness of customers to purchase its technology, competitive factors, changes resulting from healthcare policy, dependence upon third-party vendors, timing of regulatory approvals, the impact of pandemics or other disasters, and other risks discussed in the Company’s periodic and other filings with the SEC. By making these forward-looking statements, the Company undertakes no obligation to update these statements for revisions or changes after the date of this press release. There can be no assurance that the Company will recognize revenue related to customer purchase orders and other commitments because some of these purchase orders and other commitments are subject to contingencies that are outside of the Company’s control and may be revised, modified, delayed, or canceled. Stereotaxis Contacts: David L. FischelChairman and Chief Executive Officer Kimberly PeeryChief Financial Officer 314-678-6100Investors@Stereotaxis.com
Author: Ken Dropiewski
New LockeT Data Accepted in Leading Electrophysiology Journal
FORT MILL, S.C., April 06, 2026 (GLOBE NEWSWIRE) — Catheter Precision, Inc. (NYSE American: VTAK) (“VTAK” or the “Company”) today announced that the manuscript titled “Safety and Efficacy of LockeT Suture Retention Device for Large-Bore Venous Access Closure” has been accepted by the Journal of Interventional Cardiac Electrophysiology (JICE), a leading medical journal in the field of electrophysiology. This publication confirmed the safety and efficacy of LockeT for large-bore venous closure in 139 patients and concluded that LockeT demonstrated high procedural success, rapid hemostasis, early ambulation, and low complication rates for venous closure following EP procedures requiring LBVA. These findings support LockeT as a feasible and effective venous closure strategy in contemporary EP practice. David Jenkins, CEO of Catheter Precision, says, “This peer-reviewed validation from JICE reinforces our position that LockeT is a beneficial product for the physicians, patients and hospitals. The acceptance of study data in a prestigious medical journal underscores the importance of the data and enables Catheter Precision to continue its mission of product innovation.” Mr. Jenkins continued, “This additional publication on LockeT technology coupled with expanded hospital interest in both the US and Europe converges into our strategy of unlocking value in this product line.” Last week VTAK announced the engagement of a strategic advisor to assist in the company of exploring various options for its distinctive electrophysiology products. About LockeTCatheter Precision’s LockeT is a suture retention device intended to assist in wound closure after percutaneous venous punctures. LockeT is a Class 1 device registered with the FDA and has received CE Mark approval. About Catheter PrecisionCatheter Precision is an innovative U.S.-based medical device company bringing new solutions to market to improve the treatment of cardiac arrhythmias. It is focused on developing groundbreaking technology for electrophysiology procedures by collaborating with physicians and continuously advancing its products. Cautionary Note Regarding Forward-Looking StatementsThis press release contains forward-looking statements within the meaning of the Private Securities Litigation Reform Act of 1995. Words such as “believe,” “expect,” “anticipate,” “potential,” “will,” “may,” and similar expressions are intended to identify forward-looking statements. These statements include, but are not limited to, expectations regarding potential strategic transactions, valuation outcomes, market opportunities, and the Company’s growth strategy. These statements involve risks and uncertainties that could cause actual results to differ materially. For a discussion of these risks, please refer to the Company’s filings with the SEC, including its most recent Forms 10-K and 10-Q. The Company undertakes no obligation to update any forward-looking statements. CONTACTS: Investor Relations973-691-2000IR@catheterprecision.com # # #
Investor Insights: Terri Burke of Intuitive Ventures – What the team is looking to see in a meeting
Embolization, Inc., Polymer-Based Coil Demonstrates Success in Limited-Market Release
— Coils minimize artifacts in imaging with high packing density — BOULDER, Colo., April 2, 2026 /PRNewswire/ — The novel, radiopaque, polymer-based embolic coil from Embolization, Inc., is demonstrating consistent success in its limited-market release, with use cases topping 70 coils….
VDYNE Receives FDA Approval to Initiate the TRIVITA1 IDE Pivotal Trial of Transcatheter Tricuspid Valve Replacement System
MAPLE GROVE, Minn.–(BUSINESS WIRE)–VDYNE, Inc. (“VDYNE” or “the Company”), a privately held medical device company developing next generation transcatheter valve replacement technologies, today announced that the U.S. Food & Drug Administration (FDA) has approved an investigational device exemption (IDE) for the company’s pivotal clinical trial evaluating its Transcatheter Tricuspid Valve […]
BioCardia Files Request for Meeting With FDA to Discuss Accelerated Approval Pathway for CardiAMP® System in Ischemic Heart Failure
SUNNYVALE, Calif., April 02, 2026 (GLOBE NEWSWIRE) — BioCardia®, Inc. [Nasdaq: BCDA], a global leader in cellular and cell-derived therapeutics for the treatment of cardiovascular and pulmonary diseases, today reported submission to United States Food and Drug Administration (FDA) of the CardiAMP HF clinical study data and on its plans to meet with the FDA to discuss the accelerated approval pathway for the CardiAMP® System for ischemic chronic heart failure with reduced ejection fraction (HFrEF). This meeting request is in line with previous guidance and BioCardia expects to have the meeting during this quarter. The meeting will be held under BioCardia’s FDA Breakthrough Designation for the CardiAMP System. The key objectives of the meeting are to obtain FDA feedback on the acceptability of BioCardia’s proposed submission for approval based on the safety seen in the CardiAMP HF Trial, the well-characterized clinical response data for the 125 ischemic HFrEF patients enrolled into the trial, and the low risk and high potential benefit profile in the subgroup with elevated biomarkers of heart stress participating in the trial. For the CardiAMP HF trial subgroup with elevated biomarkers of heart stress, the cells delivered have shown meaningful benefit for these higher-risk patients already on stable American Heart Association and American College of Cardiology guideline directed medical therapy. Demonstrated benefits in the subgroup, in addition to benefits the patients received from guideline directed medical therapy over two years, include an additional 47% relative risk reduction in all cause cardiac death, 37% relative risk reduction in non-fatal major adverse cardiac events (stroke, myocardial infarction, and hospitalization), and meaningful improvements in quality of life (p=0.04). The latest results from this study were presented at the THT (Technology and Heart Failure Therapeutics) 2026 Annual Meeting and are available on the BioCardia website. About CardiAMP Autologous Cell TherapyGranted FDA Breakthrough designation, CardiAMP Cell Therapy uses a patient’s own bone marrow cells delivered to the heart in a minimally invasive, catheter-based procedure intended to increase capillary density and reduce tissue fibrosis of myocardial tissue to address microvascular dysfunction. Clinical development of the CardiAMP Cell Therapy for heart failure is supported by the Maryland Stem Cell Research Fund and is reimbursed by Centers for Medicare and Medicaid Services (CMS). CAUTION – Limited by United States law to investigational use. About BioCardia® BioCardia, Inc., headquartered in Sunnyvale, California, is a global leader in cellular and cell-derived therapeutics for the treatment of cardiovascular and pulmonary disease. CardiAMP® autologous and CardiALLO™ allogeneic cell therapies are the Company’s biotherapeutic platforms with three cardiac clinical stage product candidates in development. These therapies are enabled by its Helix™ biotherapeutic delivery and Morph® vascular navigation product platforms, and soon the Heart3D™ fusion imaging platform. BioCardia selectively partners on biotherapeutic delivery with peers developing important biologic therapies. For more information visit www.biocardia.com. Forward Looking Statements:This press release contains forward-looking statements that are subject to many risks and uncertainties. Forward-looking statements include, among other things, statements relating to FDA approval of CardiAMP, the planned meeting and the likelihood of safety and patient benefit. These forward-looking statements are made as of the date of this press release. We may use terms such as “believes,” “estimates,” “anticipates,” “expects,” “plans,” “intends,” “may,” “could,” “might,” “will,” “should,” “approximately” or other words that convey the uncertainty of future events or outcomes to identify these forward-looking statements. Although we believe that we have a reasonable basis for each forward-looking statement contained herein, we caution you that forward-looking statements are not guarantees of future performance and that our actual results may differ materially from the forward-looking statements contained in this press release. Factors that could cause or contribute to such differences include, but are not limited to, the Company’s liquidity position and its ability to raise additional funds, as well as the Company’s ability to successfully progress its clinical trials. As a result of these factors, we cannot assure you that the forward-looking statements in this press release will prove to be accurate. Additional factors that could materially affect actual results can be found in BioCardia’s Form 10-K filed with the Securities and Exchange Commission on March 24, 2026, under the caption titled “Risk Factors” and in its subsequently filed Quarterly Reports on Form 10-Q. BioCardia expressly disclaims any intent or obligation to update these forward-looking statements, except as required by law. Media Contact:Miranda Peto, Marketing / Investor RelationsEmail: mpeto@BioCardia.comPhone: 650-226-0120 Investor Contact:David McClung, Chief Financial OfficerEmail: investors@BioCardia.comPhone: 650-226-0120
Catalyst MedTech Announces Acquisition of X3D
Expands OEM Platform and Strengthens Leadership in Nuclear Medicine ImagingPITTSBURGH, April 02, 2026 (GLOBE NEWSWIRE) — Catalyst MedTech, a national leader in nuclear medicine and molecular imaging solutions, today announced the acquisition of X3D ApS (“X3D”), the OEM behind widely deployed gamma camera and SPECT systems used in nuclear cardiology and general nuclear medicine. The acquisition builds on Catalyst MedTech’s Digirad portfolio of systems—Ergo, Cardius2 XPO, Cardius3 XPO, and X-ACT+. X3D, formerly DDD-Diagnostic, is a globally recognized engineering and manufacturing partner under private label to leading medical equipment companies, with approximately 5,000 systems installed worldwide. Known for precision engineering, Danish design, and system architecture, X3D has established itself as a trusted force in nuclear medicine and molecular imaging. X3D’s flagship cardiac imaging system, CorCam, is recognized for its diagnostic accuracy, operational efficiency, and compact design. Catalyst MedTech’s acquisition also introduces QuantumCam, a general-purpose gamma camera designed to offer a flexible, space-efficient, and cost-effective solution for hospitals and imaging centers seeking to modernize or expand nuclear medicine capabilities. The combination of Catalyst MedTech’s Digirad portfolio and X3D’s CorCam and QuantumCam creates one of the most comprehensive, versatile offerings in nuclear cardiology and general nuclear medicine. The expanded platform enables Catalyst MedTech to support a broader range of clinical settings while driving innovation across cardiac imaging systems, software, and clinical applications. “This acquisition marks a pivotal step in our strategy to expand access while taking greater ownership of the technologies that power nuclear medicine,” said Martin Shirley, Catalyst MedTech’s President and CEO. “By combining the proven Digirad platform with X3D’s engineering capabilities and global OEM heritage, we are uniquely positioned to accelerate technology, strengthen our market leadership, and ensure long-term support and continuity for thousands of systems already in the field.” Manufacturing of CorCam will transition to the United States, enhancing domestic production capacity and supply chain resilience. X3D’s Denmark-based engineering team will remain in place, continuing to manufacture QuantumCam systems and serving as a key hub for innovation and system development. “We’re proud that our entire organization in Denmark will continue as an integral part of the new Catalyst MedTech structure,” said Admin Ajkunic, X3D’s CEO. “This ensures continuity for our customers and partners, while reinforcing Denmark’s role as a center for innovation and advanced engineering.” About Catalyst MedTech Catalyst MedTech is a national leader in nuclear medicine and molecular imaging, delivering equipment, service, and clinical solutions to healthcare providers across the United States. Our purpose is to provide access to advanced diagnostic imaging so that patients get the care and treatment they need. As both an OEM innovator and ISO-certified service provider, Catalyst offers a comprehensive, multi-vendor approach that enables healthcare organizations to overcome barriers and expand access to advanced diagnostic imaging. The company supports cardiology, neurology, and oncology through advanced imaging technologies, including SPECT, PET, PET/CT, and optimized Brain PET. With one of the largest multi-vendor service networks in the country and a nationwide team of clinical and technical experts, Catalyst ensures imaging programs operate efficiently, reliably, and at scale. See What’s Possible.www.catalystmedtech.com Media Contact: Kate KinsellDirector of MarketingCatalyst MedTechkkinsell@catalystmedtech.com
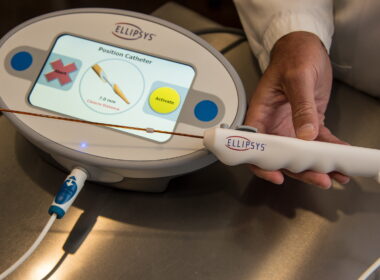
Ellipsys Vascular Access System to Return to U.S. Market in 2026
Proven percutaneous AV fistula technology set to re-enter the market with phased rollout in 2026 SAN CLEMENTE, Calif., April 2, 2026 /PRNewswire/ — Ellipsys Medical, Inc. today announced that the Ellipsys® Vascular Access System will once again be commercially available, with initial…
Catheter Precision Engages Strategic Advisor
Goal to unlock value in cardiac electrophysiology assets Strategic process to include outreach to leading global medical device companiesFORT MILL, S.C., April 02, 2026 (GLOBE NEWSWIRE) — Catheter Precision, Inc. (NYSE American: VTAK) (“VTAK” or the “Company”) today announced that it has engaged a strategic advisor to formally market for sale its cardiac electrophysiology business. This strategic initiative is designed to create shareholder value by monetizing the Company’s legacy medical device assets while sharpening its focus on the high-growth Flyte aviation platform. David Jenkins, CEO of VTAK, commented, “We believe that our cardiac electrophysiology portfolio may provide significant value to larger, better capitalized, companies than our own that is not reflected in our own company’s current market capitalization. Both LockeT and VIVO are being used by surgical and recovery teams in many hospitals in the U.S. and internationally including leading teaching hospitals. We believe LockeT provides multiple competitive advantages including large-bore closure capability, cross-functional applications spanning electrophysiology, structural heart, and interventional radiology, and lower cost per procedure. Both our product lines of LockeT and VIVO, our ventricular mapping product, have now gained FDA market clearance as well as the CE mark. With the infrastructure of a larger strategic acquirer, these assets have the potential to have their usage significantly increase within the broader cardiovascular industry. We look forward to the potential value that this strategic process may bring to our shareholders.” Mr. Jenkins continued, “By separating and monetizing these assets, we believe that over time our company will become valued in the stock market as a pure-play aviation business. Our objective is straightforward- drive shareholder value through execution, transparency, and disciplined capital allocation.” About LockeTCatheter Precision’s LockeT is a suture retention device intended to assist in wound closure after percutaneous venous punctures. LockeT is a Class 1 device registered with the FDA and has received CE Mark approval. About VIVO™ Catheter Precision’s VIVO (View Into Ventricular Onset), is a non-invasive 3D imaging system that enables physicians to identify the origin of ventricular arrhythmias pre-procedure, thereby streamlining workflow and reducing procedure time. VIVO has received marketing clearance from the U.S. FDA and has the CE Mark. About Catheter PrecisionCatheter Precision is an innovative U.S.-based medical device company bringing new solutions to market to improve the treatment of cardiac arrhythmias. It is focused on developing groundbreaking technology for electrophysiology procedures by collaborating with physicians and continuously advancing its products. Cautionary Note Regarding Forward-Looking StatementsThis press release contains forward-looking statements within the meaning of the Private Securities Litigation Reform Act of 1995. Words such as “believe,” “expect,” “anticipate,” “potential,” “will,” “may,” and similar expressions are intended to identify forward-looking statements. These statements include, but are not limited to, expectations regarding potential strategic transactions, valuation outcomes, market opportunities, and the Company’s growth strategy. These statements involve risks and uncertainties that could cause actual results to differ materially. For a discussion of these risks, please refer to the Company’s filings with the SEC, including its most recent Forms 10-K and 10-Q. The Company undertakes no obligation to update any forward-looking statements. CONTACTS: Investor Relations973-691-2000IR@catheterprecision.com # # #
Exactice Medical and ImSonic Announce Product Development and Supply Agreement for AIM Device in Transseptal Procedures
SAN DIEGO–(BUSINESS WIRE)–Exactice Medical, Inc. today announced a definitive agreement with ImSonic Medical, Inc. to complete advanced prototype development of its AIM device—the industry’s first combined imaging and transseptal access solution, designed to make transseptal procedures faster, safer, and more cost-effective. Exactice’s proprietary AIM device combines 2D/3D intracardiac echocardiography (ICE) with a fully integrated, deployable steerable needle guide. The needle guide remains