Mehran begins a one-year term leading premiere global cardiovascular WASHINGTON, March 30, 2026 /PRNewswire/ — Roxana Mehran, MD, FACC, today assumed the role of president of the American College of Cardiology. Mehran will serve a one-year term at the helm of the almost 60,000-member…
Author: Ken Dropiewski
Haemonetics Receives FDA Approval for Expanded Labeling of the VASCADE MVP® XL Venous Vascular Closure System
VASCADE MVP XL now approved for larger sheaths used in market-leading PFA and LAAC technologies BOSTON, March 30, 2026 /PRNewswire/ — Haemonetics Corporation (NYSE: HAE), a global medical technology company focused on delivering innovative solutions designed to improve patient outcomes,…
SS Innovations Expands Global Opportunity with Regulatory Approval of the SSi Mantra Surgical Robotic System in Colombia, Oman, Sri Lanka and Kenya
FORT LAUDERDALE, Fla., March 30, 2026 (GLOBE NEWSWIRE) — SS Innovations International, Inc. (the “Company” or “SS Innovations”) (Nasdaq: SSII), a developer of innovative surgical robotic technologies dedicated to making robotic surgery affordable and accessible to a global population, today announced that the Company has received regulatory approval for the SSi Mantra surgical robotic system (the “SSi Mantra”), for multiple indications in Colombia, Oman, Sri Lanka and Kenya. The Company received regulatory approval for the SSi Mantra from the Instituto Nacional de Vigilancia de Medicamentos y Alimentos (“INVIMA”) in Colombia in November 2025, from the Directorate General of Pharmaceutical Affairs and Drug Control (“DGPA&DC”) in Oman in November 2025, from the National Medicines Regulatory Authority (“NMRA”) in Sri Lanka in January 2026, and from the Pharmacy and Poisons Board (“PPB”) in Kenya in January 2026. SS Innovations believes that it is favorably positioned to address these underpenetrated markets given the SSi Mantra’s cost advantages combined with its advanced technology, differentiated features, ease of training, and user-friendliness. Dr. Sudhir Srivastava, Chairman of the Board and Chief Executive Officer of SS Innovations, commented, “We continue to expand SS Innovations’ global opportunity with an eye on democratizing access to cutting-edge robotic surgery for patients in underserved regions of the world. Over the past few months, our advanced, cost-efficient SSi Mantra surgical robotic system has received regulatory approval in Colombia, Oman, Sri Lanka, and Kenya. We are committed to decentralizing excellence in surgical robotic care in these new jurisdictions, among others, while pursuing the established United States and Europe Union markets. We anticipate that the U.S. Food and Drug Administration will complete its review of our 510(k) premarket notification for the SSi Mantra by mid-2026. Separately, we continue along the pathway towards a European Union CE marking certification for the SSi Mantra, which we believe we can obtain in 2026.” To date, the SSi Mantra has been granted regulatory approval in eleven countries, including India and the following: Colombia Oman Ecuador Philippines Guatemala Sri Lanka Indonesia Ukraine Kenya United Arab Emirates As of December 31, 2025, the SSi Mantra cumulative installed base totaled 168 across ten countries and cumulative surgeries reached 7,885, including 390 cardiac procedures and 121 pediatric surgeries. To date, more than 150 telesurgeries have been performed with the SSi Mantra. About SS Innovations SS Innovations International, Inc. (Nasdaq: SSII) develops innovative surgical robotic technologies with a vision to make the benefits of robotic surgery affordable and accessible to a larger segment of the global population. The Company’s product range includes its proprietary “SSi Mantra” surgical robotic system and its comprehensive suite of “SSi Mudra” surgical instruments, which support a variety of robotic surgical procedures including cardiac surgery. An American company headquartered in India, SS Innovations plans to expand the global presence of its technologically advanced, user-friendly, and cost-effective surgical robotic solutions. Visit the Company’s website at ssinnovations.com or LinkedIn for more information and updates. About the SSi Mantra The SSi Mantra surgical robotic system is a user-friendly, modular, multi-arm system with advanced technology features, including: 3 to 5 modular robotic arms, an open-faced ergonomic surgeon command center, a large 3D 4K monitor, a touch panel monitor for all patient related information display, a virtual real-time image of the robotic patient side arm carts, and the ability for superimposition of 3D models of diagnostic imaging. The optional SSi MantrAsana Tele Surgeon Console is a portable, compact alternative to the SSi Mantra’s standard surgeon command center that provides equivalent control functionality while enabling enhanced portability, ergonomic flexibility, and telesurgery capability. The SSi Mantra utilizes over 40 different types of robotic endo-surgical instruments to support different specialties, including cardiac surgery, and 5mm instruments for the pediatric population and ENT surgeries. A vision cart provides the table-side team with the same magnified 3D 4K view as the surgeon to provide better safety and efficiency. The SSi Mantra has been clinically validated in India in more than 100 different types of surgical procedures. Forward Looking StatementsThis press release may contain statements that are not historical facts and are considered forward-looking within the meaning of the Private Securities Litigation Reform Act of 1995. The words “anticipate,” “assume,” “believe,” “estimate,” “expect,” “will,” “intend,” “may,” “plan,” “project,” “should,” “could,” “seek,” “designed,” “potential,” “forecast,” “target,” “objective,” “goal,” or the negatives of such terms or other similar expressions to identify such forward-looking statements. These statements relate to future events or SS Innovations’ future financial performance and involve known and unknown risks, uncertainties and other factors that may cause our actual results, levels of activity, performance, or achievements to be materially different from any future results, levels of activity, performance or achievements expressed or implied by these forward-looking statements. Investor Contact:The Equity GroupKalle Ahl, CFAT: (303) 953-9878kahl@theequitygroup.com Devin Sullivan, Managing Director T: (212) 836-9608dsullivan@theequitygroup.com Media Contact:RooneyPartners LLCKate BarretteT: (212) 223-0561kbarrette@rooneypartners.com
Emboline Announces Positive Pivotal PROTECT H2H Trial Results for Emboliner® Presented as a Late-Breaking Clinical Trial at ACC 2026
Randomized IDE study met all primary and secondary safety and efficacy endpoints and demonstrated superior debris capture and significantly higher technical success versus the leading embolic protection control device in TAVR SANTA CRUZ, Calif., March 30, 2026 /PRNewswire/ — Emboline,…
Medical AI Named First-Ever Recipient of ACC Global Digital Health Award at American College of Cardiology Annual Scientific Session
AI platform recognized for real-world clinical impact through analysis of raw 12-lead ECG data, enabling earlier detection of multiple cardiovascular diseasesNEW ORLEANS, March 29, 2026 (GLOBE NEWSWIRE) — Medical AI, a company specializing in artificial intelligence-enabled electrocardiogram (ECG) solutions for cardiovascular disease detection and monitoring, today announced that it has received the inaugural Global Digital Health Award at the American College of Cardiology’s (ACC) Annual Scientific Session. The award was presented by Dr. Ami Bhatt, Chief Innovation Officer of the ACC, during the conference’s Future Hub Theater session, which highlights breakthrough technologies shaping the future of cardiovascular medicine. Medical AI was selected for its AI platform that analyzes raw data from standard 12-lead electrocardiograms, enabling clinicians to detect multiple cardiovascular conditions earlier and more accurately, including heart failure, myocardial infarction, and aortic stenosis. The award recognizes organizations outside the United States that are demonstrating measurable real-world impact through digital health innovation, including artificial intelligence, data analytics, and connected health technologies that advance cardiovascular care. “Receiving the first Global Digital Health Award from the American College of Cardiology is a tremendous honor,” said Joon-myoung Kwon, Chief Executive Officer of Medical AI. “Our goal has always been to translate advances in artificial intelligence into practical tools that help clinicians detect cardiovascular disease earlier and manage patients more effectively. This recognition underscores the growing importance of AI-driven diagnostics in improving patient outcomes around the world.” “ACC is a supporter of emerging technologies that help to further the College’s mission to transform cardiovascular care and improve heart health for all,” Dr. Bhatt said. “Medical AI’s groundbreaking work in AI to improve the cardiovascular field and lives of patients is the impactful solution we aimed to recognize with the Global Digital Health Award. Congratulations to Medical AI for this recognition.” Medical AI’s technology analyzes raw waveform data from standard 12-lead electrocardiograms to enable earlier detection and monitoring of a wide range of cardiovascular conditions, helping clinicians identify disease earlier in routine care settings. The company’s solutions have demonstrated strong real-world clinical adoption, currently deployed in 250 hospitals and health screening centers worldwide. Following national reimbursement approval in 2023, the technology is now used to evaluate approximately 220,000 patients per month on a reimbursed basis. The company’s technology platform is supported by a growing body of clinical research, including more than 70 publications in SCI-indexed journals, with findings presented in Late-Breaking Research at the 2025 European Society of Cardiology Heart Failure Congress. Medical AI has received regulatory approvals in six countries, including CE marking, and is currently pursuing U.S. Food and Drug Administration clearance, expected later this year. Dr. Hak Seung Lee, Chief Medical Officer of Medical AI, who accepted the award on behalf of the company, said, “Artificial intelligence has the potential to fundamentally reshape how cardiovascular disease is detected and managed. By extracting deeper insights from standard ECG tests, we can help physicians identify signs of cardiovascular disease earlier and make more informed decisions in everyday clinical settings. We are pleased that ACC has recognized the importance of the work we are doing with this award.” The American College of Cardiology Annual Scientific Session is one of the world’s leading gatherings focused on cardiovascular medicine, bringing together cardiologists, researchers, and healthcare innovators from across the globe to share the latest advances in science, technology, and clinical practice. About Medical AI Medical AI is a digital health company focused on developing artificial intelligence technologies that support clinicians in the diagnosis and management of cardiovascular disease. The company specializes in AI-enabled electrocardiogram analysis solutions that leverage raw ECG data to enable earlier detection of multiple cardiovascular conditions and support more accurate clinical decision-making at scale across diverse care settings. The company’s technology platform is supported by a growing body of clinical research, including more than 70 SCI-indexed publications. Medical AI has obtained regulatory approvals in six countries, including CE marking, and is currently pursuing U.S. Food and Drug Administration clearance, expected in 2026. Medical AI is headquartered in Seoul. For more information, visit www.medicalai.com. CONTACT: U.S. Media Contact:
Eliza Schleifstein
ES Media
+1 917 763-8106
eliza@schleifsteinpr.com
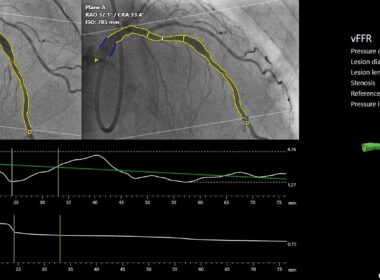
FAST III trial demonstrates non-inferiority of CAAS vFFR to invasive wire-based FFR
FAST III was initiated five years ago to evaluate whether revascularization guided by angiography–based FFR using CAAS vFFR (vessel Fractional Flow Reserve) delivers therapeutic outcomes comparable to those achieved with pressure wire–based FFR. Positive non-inferior result validates…
REPATHA® CUTS RISK OF FIRST MAJOR CARDIOVASCULAR EVENTS BY 31% IN HIGH-RISK PATIENTS WITHOUT KNOWN SIGNIFICANT ATHEROSCLEROSIS
Repatha is the Only PCSK9 Inhibitor to Significantly Reduce the Risk of First CV Events in High-Risk Primary Prevention VESALIUS-CV Subgroup Findings Reinforce Benefit of Earlier Initiation of Repatha in High-Risk Patients, with Median 44 mg/dL LDL-C Achieved THOUSAND OAKS, Calif., March…
CHAMPION-AF study of the WATCHMAN FLX™ Left Atrial Appendage Closure Device as a first-line therapy for stroke risk reduction meets all primary and secondary safety and efficacy endpoints
Data highlights the WATCHMAN FLX device provided statistically superior protection from bleeding, demonstrated similar efficacy compared to blood thinners in patients with non-valvular atrial fibrillation Late-breaking findings presented at ACC.26 and simultaneously published in The New…
Anumana Secures FDA Clearance for First-of-Its-Kind ECG-AI Algorithm for Early Detection of Pulmonary Hypertension
CAMBRIDGE, Mass.–(BUSINESS WIRE)–Anumana has received U.S. Food and Drug Administration (FDA) 510(k) clearance for its pulmonary hypertension (PH) algorithm.
HI-PEITHO trial demonstrates Boston Scientific EKOS™ Endovascular System is superior to standard of care for treatment of acute pulmonary embolism
Global randomized trial demonstrated statistically significant reduction in clinical event rates in patients with intermediate-risk PE when treated with the EKOS device plus anticoagulation vs. anticoagulation alone Late breaking findings presented at ACC.26 and simultaneously published…