Frauenfeld, Switzerland – February 2nd, 2023 SIS Medical AG (Swiss Interventional Systems), a globally leading developer, manufacturer and distributor of high-quality and high-performance products in the field of interventional cardiology, announces the launch of its lead product OPN NC® PTCA Dilatation Catheter with TWIN-Wall technology in the US. The product […]
Author: Ken Dropiewski
ProterixBio Announces Commercial Offering of Quantitative Soluble ST2 Assay
BILLERICA, Mass., Feb. 10, 2023 /PRNewswire/ — ProterixBio, Inc. today announced the offering of the Presage® ST2 Assay as a testing service through ProterixBio’s high-complexity CLIA laboratory. This Assay has been FDA cleared for use in conjunction with clinical evaluation as an aid in assessing the prognosis of patients diagnosed with chronic heart failure. Heart […]
VST-BIO Announces Successful Preclinical Trials in Ischemic Stroke
VST-Bio’s Collaborators Present Preclinical Trial Results at the American Heart Association International Stroke Conference in Dallas. SAN DIEGO, Feb. 10, 2023 /PRNewswire/ — VST-Bio Corp. a leader in the development of innovative biologics to treat acute and chronic cardiovascular disease, presented data from a recent large animal study performed by VST-Bio and Yale University demonstrating […]
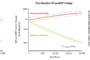
Latest Data Demonstrate Potential of the Swoop® Portable MR Imaging® System in the Follow-up of Stroke Patients Who Have Received a Thrombectomy
New study utilizes Hyperfine, Inc. portable MRI system in monitoring brain injury and evaluating physiological impacts GUILFORD, Conn., Feb. 09, 2023 (GLOBE NEWSWIRE) — Hyperfine, Inc. (Nasdaq: HYPR), the groundbreaking medical device company that created the Swoop® system, the world’s first FDA-cleared portable MRI system, today announced the presentation of new […]
Heartseed and Novo Nordisk announce first patient dosed in clinical study with HS-001 – a cell therapy designed to restore heart function in people with advanced heart failure
HS-001 consists of clusters of purified heart muscle cells derived from induced pluripotent stem cells (iPSCs). Clinical phase 1/2 study (LAPiS Study) will enrol 10 patients with advanced heart failure caused by ischaemic heart disease. Tokyo, Japan and Bagsværd, Denmark, 10 February 2023 – Heartseed Inc. (Heartseed) and Novo Nordisk A/S today announced that Heartseed has successfully dosed the first patient in […]
Syntach and Meditrial announce partnership covering clinical development of Cardiac Support System
LUND, Sweden, Feb 9, 2023 /PRNewswire/ – Syntach AB (“Syntach”) is pleased to announce that it has entered into a partnership agreement with Meditrial Europe Ltd (“Meditrial”) to support the clinical and regulatory strategies of Syntach’s innovative minimally invasive cardiac support system. Over 64 million people suffer from heart failure, one of […]
ABBOTT TO ACQUIRE CARDIOVASCULAR SYSTEMS, INC.
Abbott will gain an innovative, complementary solution in treating vascular disease through CSI’s leading atherectomy system, which prepares vessels for angioplasty or stenting to restore blood flow CSI’s offering will support Abbott’s ability to provide better care for patients with peripheral and coronary artery disease ABBOTT PARK, Ill. and ST. PAUL, […]
Philips and TriHealth announce multi-year partnership at heart of new TriHealth Heart & Vascular Institute on the campus of Bethesda North Hospital
With access to innovative healthcare technologies, clinicians can continue to provide exceptional care to local and surrounding communities and deliver on Quadruple Aim Amsterdam, the Netherlands and Cincinnati, Ohio, USA – Royal Philips (NYSE: PHG, AEX: PHIA), a global leader in health technology, has entered into a long-term strategic partnership with TriHealth, […]
BioAro is the first Canadian company to launch comprehensive genetic testing panels for various heart diseases
CALGARY, AB, Feb. 9, 2023 /PRNewswire/ – Cardiovascular diseases affects over 3 million Canadians, and over 750, 000 Canadians are living with heart failure. Almost 1 in 3 Canadians are touched by heart failure, and over 100,000 new Canadians are diagnosed with heart failure every year. The death rate is 3 times higher […]
AbbVie Reports Full-Year and Fourth-Quarter 2022 Financial Results
Reports Full-Year Diluted EPS of $6.63 on a GAAP Basis, an Increase of 2.8 Percent; Adjusted Diluted EPS of $13.77, an Increase of 16.4 Percent; These Results Include an Unfavorable Impact of $0.39 Per Share related to 2022 Acquired IPR&D and Milestones Expense1 Delivers Full-Year Net Revenues of $58.054 Billion, an Increase of 3.3 Percent on […]