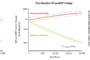
The OCEANIC program will investigate the efficacy and safety of asundexian (BAY2433334) in the prevention of stroke in patients with atrial fibrillation and also patients with a non-cardioembolic ischemic stroke or high-risk transient ischemic attack (TIA)1,2 OCEANIC-AF and OCEANIC-STROKE are expected to enroll more than 27,000 patients in over 40 […]
Author: Ken Dropiewski
LivaNova Initiates Limited Commercial Release in Europe of the Essenz Perfusion System for Cardiopulmonary Bypass Procedures
LONDON–(BUSINESS WIRE)–LivaNova PLC (Nasdaq: LIVN), a market-leading medical technology and innovation company, today announced the start of a limited commercial release for the Essenz™ Perfusion System*. The release has been initiated in select centers throughout Europe, following a successful clinical experience in two major centers, Catharina Hospital in Eindhoven, Netherlands […]
Rhythm Management Group Awarded National Remote Monitoring Services and Solutions Agreement with Premier, Inc.
ROCKVILLE, Md., Feb. 7, 2023 /PRNewswire/ — Rhythm Management Group (Rhythm), a market leading service provider of remote monitoring services, has been awarded a national group purchasing agreement for Remote Patient Monitoring of Cardiac Implantable Electronic Devices (CIEDs) and external physiologic equipment and devices with Premier, Inc., a leading healthcare improvement company headquartered in Charlotte, North Carolina. Effective […]
Northeast Scientific Inc. awarded 510(k) clearance to reprocess the Eagle Eye Platinum RX Digital IVUS Catheter
WATERBURY, Conn., Feb. 06, 2023 (GLOBE NEWSWIRE) — Northeast Scientific Inc., the pioneers in reprocessing single use peripheral vascular catheters, announced this week it has received FDA 510(k) clearance for reprocessing the Philips IVUS Eagle Eye Platinum RX Digital catheter. CEO and Founder, Craig Allmendinger shared his thoughts on the […]
Orchestra BioMed Strengthens Senior Leadership Team
Avi Fischer, M.D., joins as SVP, Medical Affairs and Innovation; J.C. Simeon assumes role of SVP, Quality George Papandreou, Ph.D., promoted to General Manager and SVP, Focal Therapies; Kunal Faldu promoted to VP, Pharmaceutical Development NEW HOPE, Pa., Feb. 07, 2023 (GLOBE NEWSWIRE) — Orchestra BioMed Holdings, Inc. (Nasdaq: OBIO, […]
BioSig Appoints Former Qorvo CFO as Successor to Steve Chaussy
Westport, CT, Feb. 07, 2023 (GLOBE NEWSWIRE) — Chief Financial Officer Steve Chaussy retires following 12 years of service Steve Buhaly joins BioSig as CFO Buhaly brings 15 years of public company CFO experience, including 9 years of leadership with RF chip technology giant Qorvo BioSig Technologies, Inc. (NASDAQ: BSGM) (“BioSig” or the […]
Sensydia Achieves Study Target for Heart-Sound AI
— Completion of enrollment for this 225-subject study is a key milestone on the path to delivering the first device for non-invasive and accurate measurement of pulmonary pressure — LOS ANGELES–(BUSINESS WIRE)–Non-invasive cardiac assessment company Sensydia announced today that it has completed its 225-subject development study at the University of Pittsburgh Medical Center (UPMC). […]
DiA Secures FDA Clearance for New AI-Powered Software, Assisting Ultrasound Users To Capture High-Quality Images
DiA Imaging Analysis’s LVivo IQS, a vendor-neutral AI-powered software provides cardiac ultrasound users with real-time image quality feedback while scanning BEER SHEVA, Israel, Feb. 6, 2023 /PRNewswire/ — DiA Imaging Analysis, a leading provider of advanced AI-based software solutions for ultrasound image analysis, announced today that the U.S. Food and Drug Administration (FDA) […]
CardieX Subsidiary ATCOR Partners with Power to Advance Patient Recruitment Diversity and Increase Patient Access in Clinical Trials
CardieX’s ATCOR subsidiary and Power together will expand patient-centric approaches to enhance patient diversity in clinical trials IRVINE, Calif., Feb. 6, 2023 /PRNewswire/ — CardieX Limited, a global health technology company focused on patient-centric healthcare research and improving clinical trial patient outcomes, today announced a partnership between its ATCOR subsidiary and Power, a fast growing patient […]
Joint Guideline on Acute Mechanical Circulatory Support Released by the Heart Failure Society of America and the International Society for Heart and Lung Transplantation
ROCKVILLE, Md., Feb. 6, 2023 /PRNewswire/ — A new joint guideline published today by the Heart Failure Society of America (HFSA) and the International Society for Heart and Lung Transplantation (ISHLT) addresses the growing availability and use of acute or temporary mechanical support devices. The joint guideline provides critical recommendations and protocols for the […]