January 31, 2023 Latest innovation has the potential to reduce anesthesia during minimally invasive procedures [1] As a pioneer and leading innovator in cardiac ultrasound, Philips has developed the smaller X11- 4t Mini 3D TEE transducer to serve more patients with this valuable imaging tool, from pediatric patients to older […]
Coronary/Structural Heart
Avenacy Announces Launch of Bivalirudin for Injection in the U.S. Market
January 29, 2024 08:00 AM Eastern Standard Time SCHAUMBURG, Ill.–(BUSINESS WIRE)–Avenacy, a specialty pharmaceutical company focused on supplying critical injectable medications, today announced it has launched Bivalirudin for Injection in the United States as a therapeutic equivalent generic for Angiomax® for Injection (bivalirudin) approved by the U.S. Food and Drug […]
Rudolf Riester GmbH Announces Global Market Launch of Telemedicine Solution, Showcases ri-sonic® E-Stethoscope + eMurmur® AI Integration for Heart Murmur Detection at Arab Health 2024
JUNGINGEN, Germany–(BUSINESS WIRE)–Rudolf Riester GmbH, a global leader in medical technology, proudly announces the full market launch of its comprehensive Telemedicine offering. Already in active use across Europe, the Middle East, and Africa, the solution from the trusted Riester brand delivers industry-leading quality and versatility through integration of a broad range of […]
PrepMD’s Online Cardiac Healthcare Training Solutions Now Approved for CEUs
BRAINTREE, Mass., Jan. 26, 2024 /PRNewswire/ — PrepMD, the leading cardiac healthcare solutions provider, announced today the accreditation of their online healthcare training programs for Nursing Continuing Education Units (CEUs). Achieved in collaboration with Gladwell CME, a pioneer…
Cytokinetics Presents New Data at CMR 2024 From FOREST-HCM, the Open Label Extension Clinical Trial of Aficamten
SOUTH SAN FRANCISCO, Calif., Jan. 25, 2024 (GLOBE NEWSWIRE) — Cytokinetics, Incorporated (Nasdaq: CYTK) today announced new data at CMR 2024 demonstrating favorable effects on cardiac structure, function and fibrosis related to treatment with aficamten in FOREST-HCM (Follow-up, Open-Label, Research Evaluation of Sustained Treatment with Aficamten in HCM), the open label extension clinical trial of aficamten in patients with hypertrophic cardiomyopathy (HCM). In many patients with […]
CroíValve Announces FDA IDE Approval and Initiation of its Early Feasibility Study
DUBLIN–(BUSINESS WIRE)–CroíValve, an Irish-U.S. based medical device company with a novel, transcatheter solution for tricuspid regurgitation (TR), today announced initiation of its Early Feasibility Study (EFS) for the DUO™ Tricuspid Coaptation Valve System, following FDA IDE and CMS approval. The TANDEM II trial is a prospective, multi-center, non-randomized single-arm study […]
HeartFlow Announces New Reimbursement Code and Increased Payment for FFRCT
MOUNTAIN VIEW, Calif., Jan. 23, 2024 (GLOBE NEWSWIRE) — HeartFlow, Inc., a leader in non-invasive artificial intelligence (AI) precision coronary solutions, announced FFRCT was transitioned to a Category I Current Procedural Terminology (CPT®) code beginning January 1, 2024. The new code, designated by The American Medical Association (AMA), supports FFRCT […]
New Survey Reveals 86 Percent of Benefits Leaders Recognize Need for Heart Health Strategy in 2024
MENLO PARK, Calif.–(BUSINESS WIRE)–Hello Heart, the digital leader in preventive heart health, today released its inaugural Why Heart Health Matters Report, which examines the toll that cardiovascular disease (CVD) takes on employers, health plans, and the people they serve. The report surveyed 300 benefits leaders at large employers and health plans […]
Pi-Cardia Receives FDA Breakthrough Device Designation for ShortCut™
REHOVOT, Israel–(BUSINESS WIRE)–Pi-Cardia Ltd., a global leader in the development of non-implant, catheter-based, leaflet modification solutions for treating heart valves, announced today it received Breakthrough Device Designation from the US Food and Drug Administration for ShortCut™ – the world’s first dedicated leaflet modification device facilitating valve-in-valve Transcatheter Aortic Valve Replacement […]
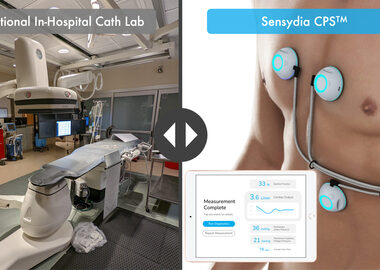
Sensydia Completes Fifth Study for Heart-Sound AI
LOS ANGELES–(BUSINESS WIRE)–Non-invasive cardiac assessment company Sensydia announced today that it has completed its 50-subject development study at the University of Minnesota (UMN). This study was conducted at UMN to collect data for its innovative AI-powered, non-invasive Cardiac Performance System (CPS™) that uses heart sound analysis to enable earlier detection and more effective therapy […]