MILFORD, Mass.–(BUSINESS WIRE)–Reprieve Cardiovascular, Inc., a development stage company focused on pioneering an intelligent automated diuretic and fluid management approach for acute decompensated heart failure (ADHF) treatment, today announced it has raised a total of $42 million in series A financing. The total round was co-led by Lightstone Ventures and […]
Coronary/Structural Heart
Cleerly® Announces CERTAIN Multicenter Clinical Trial Results
Study Results Demonstrate the Prominent Effects of Cleerly’s Products on Changing Clinical Management for Patients Suspected of Coronary Artery Disease DENVER – February 8, 2024 — Cleerly, the company working to create a new standard of care to aid in the diagnosis of heart disease, announced continued strong scientific evidence supporting […]
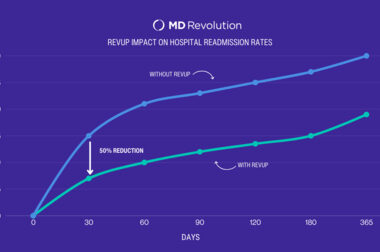
Study Results: MD Revolution and Cardiac Solutions Achieve 50% Reduction in 30-Day Cardiovascular Readmissions Using RevUp Remote Care Management
SAN DIEGO–(BUSINESS WIRE)–MD Revolution, the leader in remote care management solutions for health systems and multi-specialty clinics, announced today that its partner, Cardiac Solutions achieved a 50% reduction in hospital cardiovascular readmission rates by using RevUp Remote Patient Monitoring and Chronic Care Management as part of their program. “Our goal […]
Genesis MedTech Completes Enrollment in its United States Early Feasibility Study Utilizing the J-Valve™ Transfemoral System for Patients with Severe Aortic Regurgitation
BURLINGAME, Calif., Feb. 5, 2024 /PRNewswire/ — Genesis MedTech, a leading medical device company, today announced the completion of enrollment in its United States Early Feasibility Study (EFS) using its dedicated transcatheter aortic valve replacement (TAVR) system, J-Valve™ Transfemoral (TF) System. Finalizing the EFS marks another significant milestone for Genesis MedTech and its U.S.-based […]
Houston-Based resTOR Longevity Clinic to Offer Cardio Diagnostics’ AI-Driven Epigenetic-Genetic Heart Disease Tests
CHICAGO–(BUSINESS WIRE)–Cardio Diagnostics Holdings, Inc. (NASDAQ: CDIO), an AI-driven precision cardiovascular medicine company, announces that Houston-based resTOR Longevity Clinic will integrate Cardio Diagnostics’ solutions into its battery of tests for new patients in its longevity-focused concierge clinic. This collaboration marks a significant milestone for Cardio Diagnostics, as resTOR will be the […]
Acarix CADScor System Now Listed on U.S. VA Federal Supply Schedule
NEW YORK, Feb. 5, 2024 /PRNewswire/ — Acarix, a leader in advanced acoustic-based cardiac diagnostic devices today announces the CADScor® System has been added to the United States Federal Supply Schedule (FSS). The nationwide contract simplifies purchasing for the Veterans Health…
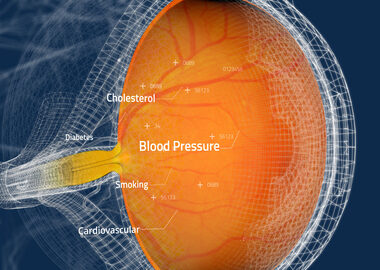
Toku Obtains CE and UKCA Marks for AI Cardiovascular Risk Assessments Through the Eye
SAN DIEGO–(BUSINESS WIRE)–Toku, Inc., a commercial medical device company specializing in AI assessment of retinal images, announced today that it has obtained CE and UKCA Marks for its patented CLAiR technology. This significant milestone underscores Toku’s commitment to maintaining the highest standards of quality and safety and allows access to […]
Edwards’ EVOQUE Valve Replacement System First Transcatheter Therapy to Earn FDA Approval for Tricuspid Valve
IRVINE, Calif.–(BUSINESS WIRE)– Edwards Lifesciences Corporation (NYSE: EW) today announced the company’s EVOQUE tricuspid valve replacement system is the first transcatheter therapy to receive U.S. Food and Drug Administration (FDA) approval for the treatment of tricuspid regurgitation (TR). The EVOQUE system is indicated for the improvement of health status in patients […]
SoniVie announces enrollment completion in the REDUCED-1 pilot study with TIVUS™ Ultra-Sound Renal Denervation
SoniVie announces enrollment completion in the REDUCED-1 pilot study TEL AVIV, Israel, Jan. 31, 2024 /PRNewswire/ — SoniVie, an Israeli company developing a novel, proprietary Therapeutic Intra-Vascular Ultrasound (TIVUSTM) System to treat a variety of hypertensive disorders, announced…
Genomics plc publishes clinical trial results demonstrating successful integration and clinical utility of integrated risk scores combining polygenic and clinical risk of cardiovascular disease in NHS primary care
Published in the European Journal of Preventive Cardiology, the paper demonstrates that polygenic risk score information can be successfully integrated into current general practitioner (GP) clinical practice for the prevention of cardiovascular disease The paper also demonstrates that…