MADRID, Jan. 28, 2026 /PRNewswire/ — The ReDS-SAFE HF II Trial, a large multicenter investigator-initiated randomized controlled study, funded by both the Instituto de Salud Carlos III and the Spanish Society of Cardiology, has officially commenced. The trial is designed to further…
Coronary/Structural Heart
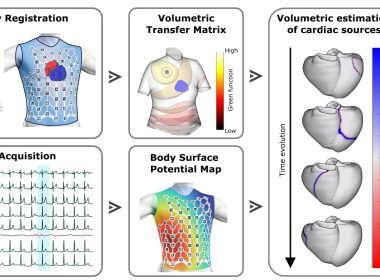
Corify Care Introduces First Whole-Heart Mapping Technology, Delivering a Global View of Cardiac Arrhythmias in a Single Beat
MADRID, Jan. 28, 2026 /PRNewswire/ — Corify Care today announced a major breakthrough in cardiac electrophysiology with the publication of its proprietary Global Volumetric Mapping technology in Nature Communications Medicine. The publication marks the first system capable of mapping all…
New Scientific Statement Guides Clinicians on How to Integrate Digital Health Tools into Heart Failure Care
WASHINGTON, Jan. 27, 2026 /PRNewswire/ — A new scientific statement provides clinicians with practical guidance on how to integrate digital health tools into everyday heart failure care – moving beyond isolated devices toward coordinated, team-based, and actionable systems of care….
Da Vinci 5 Cleared for Cardiac Procedures
FDA 510(k) clearance expands da Vinci 5 indications to include certain cardiac procedures FDA 510(k) clearance expands da Vinci 5 indications to include certain cardiac procedures
Concept Medical Announces First Patient Enrollment in STARS DAPT Randomized Clinical Trial Evaluating Abluminus NP Polymer-Free DES
TAMPA, Fla.–(BUSINESS WIRE)–Concept Medical Inc., a global pioneer in innovative drug delivery technology, is proud to announce the successful enrollment of the first patients in the STARS DAPT (ST-Segment Elevation Myocardial InfArction treated with a Polymer-Free Sirolimus-based NanocarrieR Eluting Stent and a P2Y12 inhibitor-based Single Antiplatelet Strategy after a Short Dual Antiplatelet Therapy […]
CVRx Announces Initiation of BENEFIT-HF, a Landmark Heart Failure Trial Evaluating Barostim in Significantly Expanded Population
Study is Expected to Be One of the Largest Therapeutic Cardiac Device Trials in Heart Failure Ever Performed, and is Supported by CMS Category B IDE CoverageMINNEAPOLIS, Jan. 22, 2026 (GLOBE NEWSWIRE) — CVRx, Inc. (NASDAQ: CVRX), a medical device company focused on developing, manufacturing and commercializing innovative neuromodulation solutions for patients with cardiovascular diseases, today announced initiation of the BENEFIT-HF clinical trial, a landmark randomized study supported by CMS Category B IDE coverage and designed to evaluate the impact of Barostim™ in a significantly expanded heart failure population. Barostim™ is an implantable device that delivers electrical signals to baroreceptors located on the carotid artery, increasing baroreflex signaling, rebalancing the autonomic nervous system, and improving heart failure symptoms. Barostim™ received FDA approval in 2019 following its designation under the FDA’s Breakthrough Devices Program and is now commercially available in both the U.S. and Europe. The BENEFIT-HF trial is expected to be one of the largest therapeutic cardiac device trials ever performed in heart failure, randomizing 2,500 patients in approximately 150 centers in the United States and Germany. The primary endpoint will be a composite of all-cause mortality and heart failure decompensation events. Enrollment is expected to begin in the first half of 2026. The BENEFIT-HF trial is designed to evaluate Barostim in patients with heart failure who: Remain symptomatic after receiving optimized guideline-directed medical and device therapies (GDMT)Have a left ventricular ejection fraction (LVEF)
January Issue of Journal of Cardiac Failure-Intersections Examines How Innovation, Advanced Mechanical Support, and Prescribing Design Are Reshaping Heart Failure Care
The third issue of the open-access journal showcases multidisciplinary heart failure research and the many intersections that shape patient care. WASHINGTON, Jan. 21, 2026 /PRNewswire/ — The January issue of Journal of Cardiac Failure-Intersections brings together new analyses and…
Viatris Expands Innovative Portfolio in Cardiovascular Diseases with the Company’s First Launch of Inpefa® (Sotagliflozin) for the Treatment of Heart Failure
Builds on Viatris’ Scientific Leadership and Commercial Legacy in Cardiovascular Diseases PITTSBURGH, Jan. 20, 2026 /PRNewswire/ — Viatris Inc. (Nasdaq: VTRS), a global healthcare company, today announced the launch of Inpefa® (sotagliflozin) in the United Arab Emirates (UAE), the first…
New Minimally Invasive Therapy at MemorialCare Heart & Vascular Institute Reduces Cardiovascular Mortality by Nearly 30% in Patients with Resistant Hypertension
MemorialCare introduces the Symplicity™ Renal Denervation procedure across its health system, targeting patients with uncontrolled high blood pressure—a therapy shown in clinical trials to reduce major cardiovascular events by 20%, stroke by 27%, and heart failure by 28%. FOUNTAIN VALLEY,…
Cardiac Biotech Solutions Activates China Production Pipeline for MyCardia AT and Moves into Final NMPA Submission Phase
Cardiac Biotech Solutions Activates China Production Pipeline for MyCardia AT and Moves into Final NMPA Submission Phase