New company will deliver blood tests based on large-scale protein measurements to people in Japan BOULDER, Colo., July 09, 2020 (GLOBE NEWSWIRE) — SomaLogic, Inc. and NEC Solution Innovators, Ltd. of Japan, a subsidiary of NEC Corporation, announced today the establishment of FonesLife Corporation, a new Tokyo-based company that will […]
Coronary/Structural Heart
Philips and BioIntelliSense form strategic collaboration to enhance remote patient monitoring for at-risk patients from the hospital into the home
July 9, 2020 Philips integrates the BioIntelliSense FDA-cleared BioSticker™ sensor as part of its remote patient monitoring solutions for patients outside the hospital Multi-parameter sensors aid monitoring across multiple chronic conditions with medical-grade vital signs for physicians to remotely track core symptoms, including COVID-19 Amsterdam, the Netherlands – Royal Philips (NYSE: PHG, […]
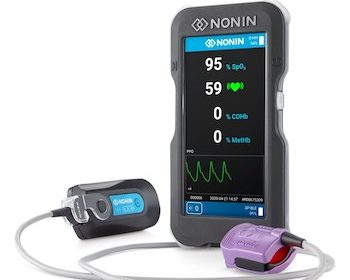
Nonin Medical Announces FDA Clearance of the CO-Pilot™ Wireless Handheld Multi-Parameter System
Device and sensors help EMS, fire departments and military track five patient parameters MINNEAPOLIS, July 8, 2020 /PRNewswire/ — Nonin Medical, Inc. today announced the CO-Pilot™ Wireless Handheld Multi-Parameter System (H500) received 510(k) clearance from the U.S. Food and Drug Administration (FDA). The CO-Pilot is a handheld device with wireless capabilities that helps […]
CHF Solutions and RenalSense Enter Distribution Partnership to Offer Ultrafiltration and Real-Time Renal Diagnostics
EDEN PRAIRIE, Minn., July 08, 2020 (GLOBE NEWSWIRE) — CHF Solutions (Nasdaq: CHFS) and RenalSense, Ltd., today announced the signing of a distribution agreement to offer complementary solutions for fluid management. The agreement allows CHF Solutions to market and sell RenalSense’s Clarity RMS™ (real-time monitoring system) in certain U.S. territories. Clarity RMS is […]
ViralClear Pharmaceuticals partners with Albany Molecular Research Inc. on the manufacture of merimepodib active pharmaceutical ingredient development for the potential treatment of COVID-19
Westport, CT, July 08, 2020 (GLOBE NEWSWIRE) — BioSig Technologies, Inc. (Nasdaq: BSGM) (“BioSig” or the “Company”) and its subsidiary, ViralClear Pharmaceuticals, Inc., today announced that it is partnering with Albany Molecular Research Inc., (AMRI), a leading global contract research, development and manufacturing organization (CDMO), for support in undertaking research to […]
SINOMED® Completes Last Patient Follow-Up of the PIONEER-III Pivotal US and Japanese Trial of the BuMA Supreme® Coronary Drug-Eluting Stent
TIANJIN, China, July 6, 2020 /PRNewswire/ — SINOMED, a developer of innovative neuro- and cardiovascular technologies, announced the completion of the 1-year follow-up in the PIONEER-III, randomized global trial evaluating the BuMA Supreme Drug-Eluting Coronary Stent (DES). Once completed, the company plans to submit the data to the U.S. Food and Drug Administration and Japanese Pharmaceuticals […]
Paragonix Technologies Announces Utilization of Paragonix SherpaPak CTS in Complex Heart Transplant Study
New data on successful transplantations for high-risk profile donor hearts presented at the American Transplant Congress CAMBRIDGE, Mass.–(BUSINESS WIRE)–Paragonix Technologies, Inc. announced the presentation of new clinical data at the American Transplant Congress (ATC) on the successful use of the Paragonix SherpaPak™ Cardiac Transport System (CTS) for high-risk donor hearts at the […]
SARS-CoV-2 Cell Entry Mechanisms in Intact Human Heart Published in JACC: Basic to Translational Science by University of Colorado Anschutz Medical Campus and ARCA biopharma Investigators
Integrin A5 may be a new target for intervening in the cell infectious process Findings may lead to the development of precision therapeutic approaches to prevent SARS-CoV-2-cell entry while preserving the functional activity of ACE2 WESTMINSTER and AURORA, Colo., July 07, 2020 (GLOBE NEWSWIRE) — The University of Colorado Anschutz Medical […]
Celixir provides update on ongoing clinical programme with lead therapeutic candidate CLXR-001
Celixir provides update on ongoing clinical programme with lead therapeutic candidate CLXR-001 First patient successfully dosed in cardiomyopathy study Stratford-upon-Avon, UK: July, 6 2019 – Celixir, a privately-owned company discovering and developing life-saving advanced therapies, announces an update to its ongoing clinical programme with lead therapeutic candidate CLXR-001. The first patient was […]
Carag Receives U.S. FDA IDE Approval to Conduct Clinical Study of First Transcatheter Septal Occluder With Bioresorbable, Metal-free Framework
The U.S. trial for CE-marked CBSO is designed to enroll up to 250 patients in a staged study approach BAAR, Switzerland, July 1, 2020 /PRNewswire/ — CARAG AG, a privately-held Swiss medical device development company, today announced receiving U.S. Food and Drug Administration (FDA) Investigational Device Exemption (IDE) approval for its Carag Bioresorbable […]