THE WOODLANDS, Texas–(BUSINESS WIRE)–Berlin Heart Inc. announced they have completed the Post Approval Surveillance for the EXCOR® Pediatric Ventricular Assist Device, a requirement of the Post Market Approval (PMA) granted by the Food and Drug Administration (FDA), in June 2017. The final report submitted to the FDA confirmed the positive contemporary […]
Coronary/Structural Heart
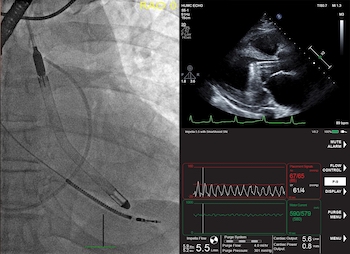
Study Finds 84% Survival Rate in Patients in Cardiogenic Shock and Other Challenging Cardiac Conditions with the New Impella 5.5 with SmartAssist
DANVERS, Mass.–(BUSINESS WIRE)–The first published United States experience of patients who received Abiomed’s newest heart pump, Impella 5.5 with SmartAssist, finds 84% of the patients survived to explant with 76% native heart recovery. The study was published in the July edition of the American Society for Artificial Internal Organs (ASAIO) Journal. The study examined […]
Esperion Announces Publication in the Journal of the American Medical Association Cardiology of Pooled Efficacy Analysis from the Phase 3 LDL-C Lowering Clinical Development Program of NEXLETOL® (bempedoic acid) Tablets
ANN ARBOR, Mich., July 01, 2020 (GLOBE NEWSWIRE) — Esperion (NASDAQ: ESPR) today announced that pooled efficacy analysis from the four Phase 3 clinical studies of NEXLETOL, an oral, once-daily LDL-cholesterol lowering medicine, was published in the Journal of the American Medical Association (JAMA) Cardiology. The four Phase 3 clinical studies […]
Matinas BioPharma Resumes Enrollment in the ENHANCE-IT and EnACT Clinical Trials
– Topline data from ENHANCE-IT study of MAT9001 vs. Vascepa® expected in Q1 2021 – – EnACT remains on track for cohort progression in Q4 2020 – BEDMINSTER, N.J., June 30, 2020 (GLOBE NEWSWIRE) — Matinas BioPharma Holdings, Inc. (NYSE AMER: MTNB), today announced that it has commenced enrollment and started dosing […]
BioCardia Announces Activation of Pivotal Trial Studying CardiAMP Cell Therapy Trial to Treat Chronic Myocardial Ischemia
First Site Initiation Visit in Trial Completed and Executive Steering Committee Named SAN CARLOS, Calif., July 01, 2020 (GLOBE NEWSWIRE) — BioCardia®, Inc. [NASDAQ:BCDA], a leader in the development of comprehensive solutions for cardiovascular regenerative therapies, today announced activation of a pivotal trial studying the Company’s investigational CardiAMP® cell therapy in the treatment of […]
scPharmaceuticals Announces FUROSCIX® NDA Resubmission
BURLINGTON, Mass.–(BUSINESS WIRE)–scPharmaceuticals Inc. (Nasdaq: SCPH), a pharmaceutical company focused on developing and commercializing products that have the potential to optimize the delivery of infused therapies, advance patient care, and reduce healthcare costs, today announced that it has resubmitted its 505(b)(2) New Drug Application (NDA) to the U.S. Food and […]
Ancora Heart Receives IDE Approval for CorCinch-HF Pivotal Study of the AccuCinch® Ventricular Restoration System
Global Study to Evaluate Percutaneous Device Designed for the Mechanical Support of the Ventricle in Heart Failure Patients SANTA CLARA, Calif.–(BUSINESS WIRE)–Ancora Heart, Inc., a company developing a novel therapy to address heart failure, today announced U.S. Food and Drug Administration (FDA) approval of its Investigational Device Exemption (IDE) application […]
CorFlow Reports Late-Breaking Data from the “MOCA I” Clinical Trial
BAAR, Switzerland–(BUSINESS WIRE)–CorFlow Therapeutics AG (“CorFlow”) announced today that Dr. Felice Gragnano, MD, Inselspital, Bern University Hospital, Bern, Switzerland, has presented interim clinical data from the MOCA I Phase 2 clinical trial during the PCR e-Course. The MOCA I study is a first-in-human (FIH) clinical trial to document the safety and feasibility of the CorFlow Controlled Flow […]
Verve Therapeutics Presents New Data in Non-Human Primates Validating Gene Editing as a Treatment Approach for Coronary Heart Disease at the ISSCR 2020 Virtual Annual Meeting
Data Demonstrate In Vivo Adenine Base Editing Can Turn Off Either PCSK9 or ANGPTL3 in the Liver and Substantially Lower LDL Cholesterol or Triglyceride Levels CAMBRIDGE, Mass.–(BUSINESS WIRE)–Verve Therapeutics, a next-generation cardiovascular company, today announced the presentation of new preclinical proof-of-concept data in non-human primates that demonstrate the successful use of base […]
Intra-Cellular Therapies Announces Positive Top-line Results from ITI-214 Phase I/II Study in Patients with Heart Failure
Study demonstrated inhibiting phosphodiesterase-1 with ITI-214 in patients with heart failure improved cardiac function by enhancing cardiac contractility and dilating systemic arteries without inducing abnormal heart rhythms. NEW YORK, June 29, 2020 (GLOBE NEWSWIRE) — Intra-Cellular Therapies, Inc. (Nasdaq:ITCI) today announced topline results from Study ITI-214-104, a Phase I/II translational study […]