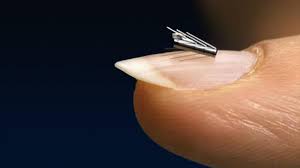
Quebec City, Quebec, April 30, 2019 – Opsens inc. (“Opsens” or the “Company”) (TSX:OPS) (OTCQX:OPSSF) is pleased to announce it has entered into a supply agreement as part of its long-term collaboration with Abiomed, Inc. (“Abiomed”) for the Impella CP® heart pump. Abiomed has awarded Opsens a five-year agreement to supply […]
Coronary/Structural Heart
New Study Finds Abbott Blood Test Can Help Predict Future Cardiac Events in Adults with No Known Heart Disease
ABBOTT PARK, Ill., April 29, 2019 /PRNewswire/ — Abbott (NYSE: ABT) announced today that a new study published in Circulation found its High Sensitive Troponin-I blood test could predict the chance of developing a cardiac event years before it occurs in people with no symptoms when added to current heart disease assessments.1 Using data from the Atherosclerosis […]
Quantum Genomics Announces the Publication of its Phase IIb NEW-HOPE Study Results of Firibastat in Arterial Hypertension in the Journal Circulation
PARIS and NEW YORK, April 26, 2019 (GLOBE NEWSWIRE) — Quantum Genomics (Euronext Growth – FR0011648971 – ALQGC), a biopharmaceutical company specializing in the development of a new drug class that directly targets the brain to treat resistant hypertension and heart failure, announces the publication of the full results from its Phase […]
New Data Confirms Cardiac Dimensions’ Carillon® System Provides Acute Hemodynamic Efficacy In Patients With Functional Mitral Regurgitation
MANNHEIM, Germany, April 26, 2019 /PRNewswire/ — Cardiac Dimensions, a leader in the development of innovative, minimally invasive treatments to address functional mitral regurgitation (FMR) in patients with heart failure (HF), presented new data confirming significant reduction in regurgitant volume directly after implantation of the Carillon Mitral Contour System® at the German Society of Cardiology […]
Neovasc Provides Highlights from Annual Conference of the DGK, the German Society of Cardiology; Symposium Generates Growing Support for Reducer
VANCOUVER, April 26, 2019 /PRNewswire/ – Neovasc Inc. (“Neovasc” or the “Company”) (NASDAQ: NVCN)(TSX: NVCN), a leader in the development of minimally invasive transcatheter mitral valve replacement technologies and in the development of minimally invasive devices for the treatment of refractory angina, today provided highlights from the 85th Annual Conference of the German […]
Paragonix Announces Transplanted-Cost Savings and Clinical Results for Donor Heart Preservation using Paragonix SherpaPak Cardiac Transport System (CTS) in Routine and Complex Heart Transplantation
CAMBRIDGE, Mass.–(BUSINESS WIRE)–Paragonix Technologies, Inc. today announced clinical results presented during the Paragonix SherpaPak™ CTS 2019 International Heart Summit held during the International Society for Heart and Lung Transplantation (ISHLT) 2019 Annual Meeting in Orlando, FL. Five U.S. and European Centers shared clinical use experiences of the Paragonix SherpaPak™ Cardiac Transport System […]
Orchestra BioMed™ Receives FDA Breakthrough Device Designation for Virtue® Sirolimus-Eluting Balloon (SEB)
NEW HOPE, Pa., April 24, 2019 (GLOBE NEWSWIRE) — Orchestra BioMed, Inc. (“Orchestra BioMed” or the “Company”), a biomedical innovation company developing advanced therapeutic solutions to address major unmet healthcare needs, announced today that it has secured Breakthrough Device designation by the U.S. Food and Drug Administration (FDA) for its Virtue® Sirolimus-Eluting […]
Boston Scientific Receives FDA Approval for LOTUS Edge™ Aortic Valve System
MARLBOROUGH, Mass., April 23, 2019 /PRNewswire/ — Boston Scientific Corporation (NYSE: BSX) today announced it has received U.S. Food and Drug Administration (FDA) approval for the LOTUS Edge™ Aortic Valve System. Delivered via a minimally-invasive procedure, this transcatheter aortic valve replacement (TAVR) technology is approved for patients with severe aortic stenosis who are considered at […]
Surmodics Sublime™ Guide Sheath Receives FDA 510(k) Approval
EDEN PRAIRIE, Minn.–(BUSINESS WIRE)–Surmodics, Inc. (NASDAQ:SRDX), a leading provider of medical device and in vitro diagnostic technologies to the health care industry, announced it has received U.S. Food and Drug Administration (FDA) 510(k) clearance for its Sublime™ guide sheath. The highly flexible, proprietary Xtreme® braid-reinforced Sublime sheath is designed to resist kinking […]
Microbot Medical Strengthens Global IP Portfolio with Canadian Notice of Allowance for a Patent Application for a Shunt Stenosis Prevention System
HINGHAM, Mass., April 23, 2019 (GLOBE NEWSWIRE) — As it continues to receive an overwhelming positive response from global jurisdictions, Microbot Medical Inc. (NASDAQ: MBOT) expands its IP portfolio as it receives a Notice of Allowance from the Canadian Intellectual Property Office for Patent Application No. 2,850,279, which pertains to […]