LONDON–(BUSINESS WIRE)–LivaNova PLC (NASDAQ: LIVN), a market-leading medical technology company, today announced the conclusion of the PRELUDE feasibility study for its Caisson Transcatheter Mitral Valve Replacement (TMVR) system. The PRELUDE first-in-human study evaluated the Company’s TMVR system to treat moderate to severe mitral regurgitation (MR) using a transseptal approach. This […]
Coronary/Structural Heart
JenaValve Technology Initiates U.S. Patient Enrollment in Early Feasibility Study of Next-Generation TAVR System for the Treatment of Severe Aortic Stenosis and Severe Aortic Regurgitation
IRVINE, Calif.–(BUSINESS WIRE)–JenaValve Technology, Inc., a developer and manufacturer of differentiated transcatheter aortic valve replacement (TAVR) systems, today announced initiation of patient enrollment in the Early Feasibility Study (EFS) of its next generation JenaValve Pericardial TAVR System using the Everdur™ transcatheter heart valve (THV) and CoronatixTMTransfemoral Delivery Catheter at NewYork-Presbyterian/ […]
Neovasc Announces Collaboration and Licensing Agreement with Penn Medicine and the Gorman Cardiovascular Research Group
VANCOUVER, Aug. 3, 2018 /PRNewswire/ – Neovasc Inc. (“Neovasc” or the “Company”) (NASDAQ: NVCN) (TSX: NVCN), a leader in the development of minimally invasive transcatheter mitral valve replacement technologies, announced today that it has entered into a collaboration and licensing agreement relating to certain know-how developed by Penn Medicine and the Gorman Cardiovascular Research […]
Janssen Receives Positive CHMP Opinion to Expand INVOKANA® and VOKANAMET® Labelling to Include Positive Data on Cardiovascular Morbidity and Mortality
BEERSE, Belgium–(BUSINESS WIRE)–The Janssen Pharmaceutical Companies of Johnson & Johnson today announced that the Committee for Medicinal Products for Human Use (CHMP), of the European Medicines Agency (EMA), has issued a positive opinion to update the INVOKANA® (canagliflozin) and VOKANAMET® (canagliflozin and metformin) labelling including changes to the indication statement for the […]
GB Sciences Co-Authors a Peer Reviewed Article Published in the Journal of the American Heart Association Titled: “Cannabinoids, The Heart of the Matter”
LAS VEGAS, Aug. 2, 2018 /PRNewswire/ — GB Sciences, Inc. (OTCQB: GBLX) is pleased to announce the publication of a review article co-authored by GB Sciences and key collaborators in the Journal of the American Heart Association, which explains the potential medical benefits of using cannabinoid-based therapies for the treatment of heart diseases. […]
Arrowhead Pharmaceuticals Earns $10 Million Milestone Payment from Amgen
PASADENA, Calif.–(BUSINESS WIRE)–Arrowhead Pharmaceuticals Inc. (NASDAQ: ARWR) today announced that it has earned a $10 million milestone payment from Amgen following the administration of the first dose of AMG 890, formerly referred to as ARO-LPA, in a clinical study. Amgen is evaluating AMG 890 in a Phase 1 clinical study […]
John Radcliffe Oxford University Hospital is the First Medical Center in the UK to Use VEST Technology in Patients Undergoing Coronary Bypass Surgery
TEL AVIV, Israel, Aug. 1, 2018 /PRNewswire/ — Vascular Graft Solutions announces today that John Radcliffe hospital in Oxford, is the first center in the UK to use its VEST technology to externally support vein grafts in patients undergoing coronary bypass surgery. “After several decades in which little progress had been made in coronary […]
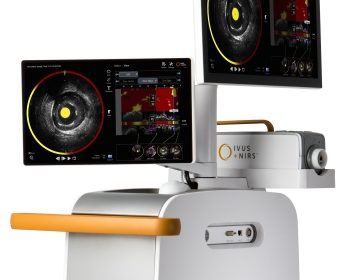
Infraredx Announces Launch of Makoto™ Intravascular Imaging System and Dualpro™ IVUS+NIRS Catheter in Japan
BURLINGTON, Mass.–(BUSINESS WIRE)–Infraredx, Inc., a pioneer in intravascular imaging for mapping coronary artery disease, today announced the Makoto™ Intravascular Imaging System, and accompanying Dualpro™ intravascular ultrasound and near-infrared spectroscopy (IVUS+NIRS) catheter, is now available in Japan. The launch follows a successful Spring 2018 limited market release, which included more than […]
GE Healthcare and Lantheus Announce Start of a Phase 3 Clinical Trial of Flurpiridaz, an Investigational Agent Being Evaluated for the Detection of Coronary Artery Disease
BOSTON & NORTH BILLERICA, Mass.–(BUSINESS WIRE)–GE Healthcare and Lantheus Holdings, Inc. (NASDAQ: LNTH), parent company of Lantheus Medical Imaging, Inc. (collectively “Lantheus”), have started a second Phase 3 clinical trial of Flurpiridaz 18F (called the AURORA study), an investigational agent being evaluated for the detection of coronary artery disease (CAD), the […]
Thermedical Announces FDA Investigational Device Exemption Approval to Begin US Clinical Study of Durablate Catheter for Treatment of Ventricular Tachycardia
WALTHAM, Mass.–(BUSINESS WIRE)–Thermedical® announced today that the U.S. Food and Drug Administration (FDA) has approved its Investigational Device Exemption (IDE) application for the Early Feasibility Study (EFS) of the groundbreaking Durablate™ ablation catheter. The single-arm, observational study is designed to evaluate the safety and effectiveness of the Durablate catheter to […]