DUBLIN and BALTIMORE – July 25, 2018 – Medtronic plc (NYSE:MDT), a global leader in heart valve therapies, will deliver public comment during the Centers for Medicare & Medicaid Services (CMS) Medicare Evidence Development and Coverage Advisory Committee (MEDCAC) meeting on July 25, which will assess whether scientific evidence supports requiring […]
Coronary/Structural Heart
New Research Demonstrates Beckman Coulter’s Access hsTnI’s Diagnostic Accuracy in Safely Ruling Out Acute Coronary Syndrome in 2 Hours
BREA, California, July 25, 2018 /PRNewswire/ — A recent article in the Annals of Emergency Medicine reported on the diagnostic accuracy of the newly released Beckman Coulter Access hsTnI assay in expediting the emergency department (ED) disposition for patients evaluated for suspected acute coronary syndrome (ACS).1 This group of experts confirmed that using serial […]
Acasti Pharma Awarded Composition-of-Matter Patent by the U.S. Patent and Trademark Office
LAVAL, Québec, July 24, 2018 (GLOBE NEWSWIRE) — Acasti Pharma Inc. (“Acasti” or the “Company”) (NASDAQ:ACST) (TSX-V:ACST), a biopharmaceutical innovator focused on the research, development and commercialization of its prescription drug candidate CaPre® (omega-3 phospholipid) for the treatment of severe hypertriglyceridemia, today announced it has received a Notice of Allowance […]
HeartStitch® Presents Initial Clinical Results Of New Tricuspid Valve Repair At CSI Frankfurt Clinical Congresses
FRANKFURT, Germany, July 24, 2018 /PRNewswire/ — HeartStitch® has presented initial clinical data of its revolutionary tricuspid valve repair technique using only surgical sutures and pledgets to repair and promote remodeling of the tricuspid valve in 30 patients suffering from tricuspid regurgitation (TR). Prof. Anthony Nobles, Chairman, CEO and Chief Clinical Officer of HeartStitch®, Inc. […]
Nation’s Top Heart Disease Patient Advocates Create Task Force Aimed at Improving Access, Research, and Awareness on Heart Valve Disease Detection and Treatment
WASHINGTON, July 24, 2018 /PRNewswire/ — The nation’s leading heart disease patient advocates announced today the launch of the Heart Valve Disease Policy Task Force, a coalition of non-profit organizations uniting for a common voice in support of improved access, research, and awareness on heart valve disease detection and treatment. The Heart Valve Disease […]
Intra-Cellular Therapies Announces Publication Highlighting ITI-214 Mechanism of Action and Describing Positive Results in Preclinical Models of Heart Failure
NEW YORK, July 23, 2018 (GLOBE NEWSWIRE) — Intra-Cellular Therapies, Inc. (Nasdaq:ITCI) today announced the online publication of a report in the journal Circulation describing the mechanism of action of its phosphodiesterase (PDE) Type 1 inhibitor, ITI-214, and its potential use for the treatment of heart failure. In these preclinical studies, Johns […]
Boston Scientific Announces Agreement to Acquire Claret Medical, Inc.
MARLBOROUGH, Mass., July 20, 2018 /PRNewswire/ — Boston Scientific Corporation (NYSE: BSX) today announced it has signed an agreement to acquire Claret Medical, Inc., a privately-held company that has developed and commercialized the Sentinel® Cerebral Embolic Protection System. The device is used to protect the brain during certain interventional procedures, predominately in patients undergoing transcatheter […]
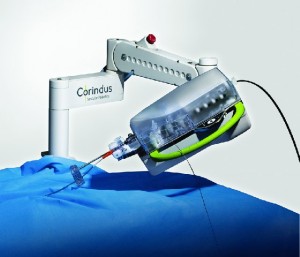
Corindus Announces Significant Progress in the Development of Remote Vascular Robotic Technology with the CorPath GRX® System
WALTHAM, Mass.–(BUSINESS WIRE)–Corindus Vascular Robotics, Inc. (NYSE American:CVRS), a leading developer of precision vascular robotics, announced today that Ryan Madder, M.D., an interventional cardiologist at Spectrum Health, has successfully completed a remote percutaneous coronary intervention (PCI) in a porcine model using the CorPath GRX System from a location greater than […]
Boehringer Ingelheim and Lilly announce Tradjenta’s CARMELINA® cardiovascular outcome trial meets primary endpoint
RIDGEFIELD, Conn. and INDIANAPOLIS, July 19, 2018 /PRNewswire/ — CARMELINA® (CArdiovascular safety and Renal Microvascular outcomE with LINAgliptin in patients with type 2 diabetes at high vascular risk) met its primary endpoint, defined as time to first occurrence of cardiovascular death, non-fatal myocardial infarction or non-fatal stroke (3-point MACE), with Tradjenta® demonstrating similar cardiovascular safety compared with […]
Fujifilm Presents Cardiovascular Imaging Solutions at the World Society for Pediatric and Congenital Heart Surgery’s 6th Scientific Meeting
STAMFORD, Conn., July 19, 2018 /PRNewswire/ — (Booth 106) — FUJIFILM Medical Systems U.S.A., Inc., a leading provider of Enterprise Imaging and Informatics solutions, will demonstrate Synapse® Cardiovascular, its comprehensive enterprise solution for cardiology, at the World Society for Pediatric and Congenital Heart Surgery’s (WSPCHS) 6thscientific meeting. This meeting is the premier gathering of the largest professional […]