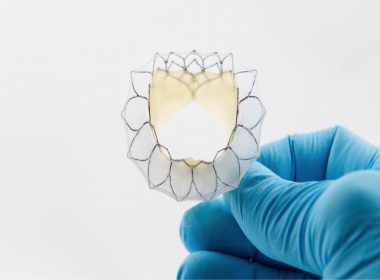
VANCOUVER, July 2, 2018 /PRNewswire/ – Neovasc Inc. (“Neovasc” or the “Company”) (NASDAQ, TZX: NVCN), a leader in the development of minimally invasive transcatheter mitral valve replacement technologies, today announced that its Tiara™ (“Tiara”), a transcatheter treatment of mitral valve disease, and the Neovasc Reducer™ (“Reducer”), a CE-Marked medical devices used for […]
Coronary/Structural Heart
CHMP issues positive opinion to expand Jardiance®, Synjardy® and Glyxambi® labels to include positive effects on cardiovascular and renal outcomes
INGELHEIM, Germany & INDIANAPOLIS–(BUSINESS WIRE)–Boehringer Ingelheim and Eli Lilly and Company (NYSE:LLY) announced today that the European Medicines Agency (EMA) Committee for Medicinal Products for Human Use (CHMP) has issued a positive opinion to update the labels of Jardiance® (empagliflozin), Synjardy® (empagliflozin and metformin) and Glyxambi® (empagliflozin and linagliptin) to include additional important […]
BioSig Technologies to Present at the Emerging Medical Technologies Spotlight
Santa Monica, CA, June 29, 2018 (GLOBE NEWSWIRE) — BioSig Technologies, Inc. (OTCQB: BSGM), a medical technology company developing a proprietary biomedical signal processing platform designed to address an unmet technology need for the $4.6 billion electrophysiology (EP) marketplace, today announced that it will be presenting at the Q2 2018 Emerging Medical […]
Broad and Bayer Expand Partnership, Launching New Research Effort to Develop Therapies for Heart Failure
CAMBRIDGE, Mass., June, 29, 2018 /PRNewswire/ — The Broad Institute of MIT and Harvard and Bayer are launching the Precision Cardiology Laboratory (PCL), a new endeavor that will pursue scientific insights aimed at developing new therapies for heart failure. According to the American Heart Association, more than 900,000 people are diagnosed with heart failure every […]
Amarin Reports Last Patient Study Visit Has Occurred, an Important Step Towards Completion of REDUCE-IT™ Cardiovascular Outcomes Study
BEDMINSTER, N.J., and DUBLIN, Ireland, June 28, 2018 (GLOBE NEWSWIRE) — Amarin Corporation plc (NASDAQ:AMRN), a biopharmaceutical company focused on the commercialization and development of therapeutics to improve cardiovascular health, today announced that the last patient study visit has occurred in its potential landmark cardiovascular outcomes study, REDUCE-IT™. The company also reiterated […]
Endotronix Joins American Heart Association’s Center for Health Technology & Innovation to Broaden the Use of Guideline-Based Care for Heart Failure Patients
LISLE, Ill., June 28, 2018 /PRNewswire/ — Endotronix, Inc., a digital health, medtech company dedicated to advancing the treatment of heart failure, today announced they have joined the American Heart Association’s Center for Health Technology & Innovation’s (CHTI) Innovators Network. The collaboration allows Endotronix to integrate the Association’s digital resources into the Cordella™ Heart […]
Edwards Comments On CMS Initiation Of National Coverage Analysis For TAVR
IRVINE, Calif., June 28, 2018 /PRNewswire/ — Edwards Lifesciences Corporation (NYSE: EW), the global leader in patient-focused innovations for structural heart disease and critical care monitoring, today commented on the announcement by the U.S. Centers for Medicare and Medicaid Services (CMS) about a National Coverage Analysis (NCA) for transcatheter aortic valve replacement (TAVR): CMS […]
Lexington Biosciences Concludes Initial HeartSentry Clinical Study
VANCOUVER, British Columbia, June 27, 2018 (GLOBE NEWSWIRE) — Lexington Biosciences, Inc. (CSE:LNB) (OTCQB:LXGTF) (the “Company” or “Lexington”), a development-stage medical device company, is pleased to announce the completion of the initial HeartSentry study conducted at San Francisco Bay-area Diablo Clinical Research. “We are pleased with the performance of the […]
Beckman Coulter Diagnostics Receives U.S. FDA 510(k) Clearance for High- sensitivity Access hsTnI Assay
BREA, Calif., June 27, 2018 /PRNewswire/ — Beckman Coulter Diagnostics announced that it has received 510(k) clearance from the U.S. Food and Drug Administration (FDA) for its new high-sensitivity troponin (hsTnI) assay, Access hsTnI, for use on the Access 2, DxI and the entire Access family of immunoassay systems. The Access hsTnI assay […]
HeartFlow Announces New Commercial Coverage With UnitedHealthcare, Largest Health Insurer in U.S.
REDWOOD CITY, Calif.–(BUSINESS WIRE)–HeartFlow, Inc. today announced that UnitedHealthcare now covers the HeartFlow® FFRct Analysis, extending access to their 45 million beneficiaries. With this new coverage, more than 235 million people in the United States now have access to the HeartFlow Analysis — significantly advancing HeartFlow’s mission of transforming cardiovascular care. The […]