BURLINGAME, Calif.–(BUSINESS WIRE)– JC Medical, Inc. announced the treatment of the first patient with the company’s Transfemoral TAVI device, the J-Valve TF System for aortic regurgitation. The first patient was treated at St. Paul’s Hospital – Vancouver, Canada by John Webb, MD, FACC, FSCAI, Director of Interventional Cardiology. There are currently […]
Coronary/Structural Heart
InspireMD Reports on Expanded 2 Year Follow-up Results from the PARADIGM Clinical Study Using CGuard EPS
TEL AVIV, ISRAEL, May 30, 2018 (GLOBE NEWSWIRE) — InspireMD, Inc. (NYSE AMER:NSPR), a leader in embolic prevention systems (EPS), thrombus management technologies and neurovascular devices, today announced that Professor Piotr Musiałek, from the Department of Cardiac and Vascular Diseases, John Paul II Hospital, Kraków, Poland, presented the expanded 24 month […]
TARA Biosystems’ CEO Misti Ushio Named to Fast Company’s Top 100 Most Creative People in Business
NEW YORK, May 30, 2018 /PRNewswire/ — TARA Biosystems Inc., a company offering physiologically relevant, human “heart-on-a-chip” tissue-models for drug discovery and development applications, is proud to announce that TARA CEO Dr. Misti Ushio has been recognized by Fast Company as one of the Top 100 Most Creative People in Business today. The annual list honors an […]
Ancora Heart Announces $17.8 Million Financing for First-of-Its-Kind Therapy for Systolic Heart Failure
SANTA CLARA, Calif.–(BUSINESS WIRE)–Ancora Heart, Inc., a company developing a novel therapy to address heart failure, today announced the closing of an initial tranche of $17.8 million in a financing round expected to reach $30 million, led by Savitr Capital and other existing investors. Ancora Heart’s AccuCinch® Ventricular Repair System is […]
ALVIMEDICA UNVEILS THE DIAB8 RANDOMIZED STUDY, THE FIRST AND ONLY DIABETIC DES RANDOMIZED TRIAL DESIGNED FOR SUPERIORITY
• Diab8 is a 55-center, 3.040-patient randomized controlled trial that pits the performance of Cre8™ EVO, a Polymer-free Amphilimus-eluting stent, against the Everolimus-Eluting stents in the treatment of coronary artery disease in diabetic patients • The Diab8 study is designed to establish the superior efficacy of the Cre8™ EVO, the […]
Intact Vascular Sponsors Symposium on Post-Angioplasty Dissections
WAYNE, Pa.–(BUSINESS WIRE)–Intact Vascular, Inc., a developer of medical devices for minimally invasive peripheral vascular procedures, today announced the schedule of key presentations that will be featured during a symposium sponsored together with Philips at the 19th Annual NCVH Conference in New Orleans on May 31. Post-Percutaneous Transluminal Angioplasty (“PTA”) dissections […]
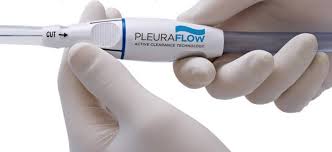
ClearFlow Announces Additional Positive US Clinical Trial Results at the 2018 NTI Critical Care Exposition
BOSTON–(BUSINESS WIRE)–ClearFlow, Inc., a medical device company based in Anaheim, California, has announced that positive clinical trial results were presented at the National Teaching Institute (NTI) & Critical Care Exposition 2018 annual meeting on May 23rd, 2018 in Boston, MA. The findings were presented by the study principal investigator, Dr. […]
GENETIC-AF Phase 2B Clinical Trial Results Presented in Late Breaking Presentation at European Society of Cardiology Heart Failure 2018 World Congress
WESTMINSTER, Colo., May 29, 2018 (GLOBE NEWSWIRE) — ARCA biopharma, Inc. (Nasdaq:ABIO), a biopharmaceutical company applying a precision medicine approach to developing genetically-targeted therapies for cardiovascular diseases, today announced that data from the GENETIC-AF clinical trial was presented in a “Late Breaking Clinical Trials” oral presentation at the European Society of Cardiology Heart Failure 2018 […]
Neovasc Provides Highlights from EuroPCR; Symposium Generates Increased Interest in Reducer
VANCOUVER, May 25, 2018 /PRNewswire/ – Neovasc Inc. (“Neovasc” or the “Company”) (NASDAQ, TSX: NVCN), a leader in the development of minimally invasive transcatheter mitral valve replacement technologies, today provided highlights from EuroPCR, the annual meeting of the European Association of Percutaneous Cardiovascular Interventions (EAPCI) of the European Society of Cardiology (ESC), which […]
Getinge announces results from SEMPER FI pilot study of intra-aortic balloon pump showing numerical trend toward reduced mortality in patients with extensive myocardial infarction and persistent ischemia
WAYNE, N.J., May 25, 2018 /PRNewswire/ — Getinge, a leading global provider of innovative medical technology, today announced results from the Survival Improvement in Extensive Myocardial Infarction with PERsistent Ischemia Following IABP Implantation (SEMPER FI) pilot study. The study evaluated the use of an intra-aortic balloon pump (IABP) in patients with extensive […]