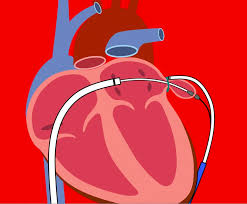
Presented at ACC.18, Five-Year Follow-up from the CoreValve Extreme Risk and NOTION Studies Show Sustained Valve Function and Durability Over Time DUBLIN and ORLANDO – March 10, 2018 – Medtronic plc (NYSE: MDT) today unveiled outcomes from the CoreValve U.S. Pivotal Extreme Risk Study and the real-world NOTION trial (Nordic Aortic […]
Coronary/Structural Heart
Lantheus Presents New Data on Novel Cardiac PET Imaging Agent LMI 1195 in Development for the Diagnostic Assessment of Ischemic Heart Failure Patients at Risk for Sudden Cardiac Death at ACC.18
NORTH BILLERICA, Mass.–(BUSINESS WIRE)–Lantheus Holdings, Inc. (NASDAQ: LNTH), the parent company of Lantheus Medical Imaging, Inc. (“LMI”), a global leader in the development, manufacture and commercialization of innovative diagnostic imaging agents, today announced positive new data from a Phase 2 study of LMI 1195, a fluorine-18 (F18) labeled tracer for […]
From Planning to Post-Op, Canon Medical Systems Delivers a Complete Suite of Structural Heart Imaging Solutions
TUSTIN, Calif.–(BUSINESS WIRE)–From accurate, precise planning to successful implementation of aortic and mitral valve replacements and post-operative evaluation, Canon Medical Systems USA, Inc. delivers a complete suite of imaging solutions for structural heart interventions. Features such as the Volumetric Scan Mode on the Aquilion™ ONE / GENESIS Edition, which enables the heart […]
SentreHEART Announces Participation at The American College of Cardiology’s 67th Annual Scientific Session & Expo
REDWOOD CITY, Calif.–(BUSINESS WIRE)–SentreHEART, Inc., the manufacturer of the LARIAT® Suture Delivery Device (LARIAT) will be participating in the American College of Cardiology (ACC) 2018 meeting being held in Orlando, Florida, March 10-12, 2018. The meeting’s agenda includes several sessions dedicated to exploring device-based options for Left Atrial Appendage (LAA) […]
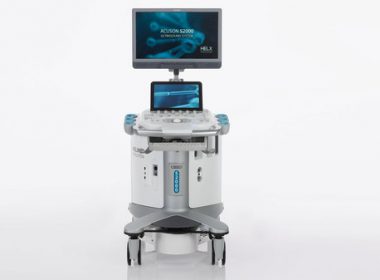
At ACC, Siemens Healthineers Introduces a Portable Cardiovascular-Dedicated Ultrasound System
ORLANDO, Fla.–(BUSINESS WIRE)– ACC 2018: Booth #3127— At the American College of Cardiology’s 67th Annual Scientific Session and Expo, Siemens Healthineers launched its new portable cardiovascular ultrasound solution, the ACUSON Bonsai. According to a recent study, 34 million echo exams were performed in the U.S. in 2017.1 The ACUSON Bonsai addresses the challenge of these […]
Marketwired: Mar 08, 2018 — Novoheart’s Landmark, Peer-Reviewed Study Published, Recognizing the Effectiveness of the World’s First and Only Human Heart-in-a-Jar
VANCOUVER, BRITISH COLUMBIA — (Marketwired) — Mar 08, 2018 — Novoheart (“Novoheart” or the “Company”) (TSX VENTURE: NVH)(FRANKFURT: 3NH) is pleased to announce that it has published a comprehensive study of the world’s first human heart-in-a-jar, in the prestigious bioengineering journal Biomaterials. The peer-reviewed paper showcases the human heart-in-a-jar’s unique ability to recapitulate […]
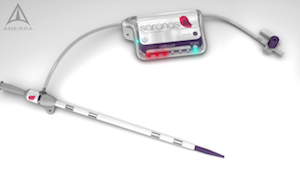
Saranas’ Novel Bleed Monitoring System Receives Recognition in Cardiovascular Research Technologies 2018 Competition
HOUSTON–(BUSINESS WIRE)– Saranas, a medical device company with a cutting-edge technology for real-time detection and monitoring of internal bleeding during endovascular procedures, placed first runner-up at the Cardiovascular Research Technologies (CRT) competition on March 6, 2018, in Washington, D.C. “We are extremely honored that our Early Bird™ Bleed Monitoring System has been recognized […]
New Prevencio Study Using AI Demonstrates Simple Blood Test Accurately Diagnoses Aortic Stenosis
KIRKLAND, Wash.–(BUSINESS WIRE)–Prevencio, Inc., today announces its HARTTM AS multi-protein, artificial intelligence (AI)-driven test, which data indicates accurately diagnoses Aortic Stenosis (AS)—a condition with a 50 percent death rate when left untreated. AS is the most common valvular heart disease in the Western world, present in more than 20 percent of […]
FDA Approves the World’s Smallest Mechanical Heart Valve for Pediatric Patients with Heart Defects
ABBOTT PARK, Ill., March 6, 2018 /PRNewswire/ — Abbott today announced the U.S. Food and Drug Administration (FDA) approved the Masters HP™ 15mm rotatable mechanical heart valve, the world’s smallest mechanical heart valve, that will allow doctors to treat babies and toddlers in need of a mitral or aortic valve replacement. Until […]
4C Medical’s Transcatheter Mitral Valve Technology Receives First Place in Cardiovascular Research Technologies 2018 Competition
BROOKLYN PARK, Minn., March 7, 2018 /PRNewswire/ — 4C Medical Technologies, Inc. (4C Medical), a developer of minimally invasive technologies for structural heart disease, today announced that AltaValve™, its medical device for mitral regurgitation (MR), was awarded first place in the Cardiovascular Research Technologies (CRT) competition held March 3-6, 2018 in Washington, DC. “AltaValve will answer […]