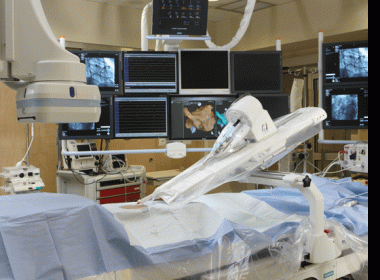
TEL AVIV, Israel, Feb. 23, 2018 /PRNewswire/ — Vascular Grafts Solutions announced today the interim results from the VEST III study, a post-marketing trial which evaluates the effectiveness of the VEST technology (Venous External SupporT) in treatment of saphenous vein graft (SVG) disease after coronary artery bypass grafting. The Interim […]
Coronary/Structural Heart
Tricuspid Valve Repair Company 4Tech Inc. Appoints Tom Fleming as President and CEO, and Dr. Keith Dawkins as CMO
GALWAY, Ireland–(BUSINESS WIRE)– 4Tech Inc., a leader in the field of transcatheter tricuspid valve repair, announces changes in its leadership team: Tom Fleming will join the company as President and Chief Executive Officer (CEO), effective as of March 2018; and Keith D. Dawkins, M.D., has joined as Chief Medical Officer (CMO), effective immediately. Tom […]
Study Validates New Approach for Optimizing Endovascular Drug Delivery Using Stents with a Deployable Coating That Enhances Sirolimus Distribution Compared to Traditional DES
“This preclinical demonstration that stent deployment of microcrystalline drug in combination with a rapidly absorbable polymer, such as with MiStent, results in more uniform drug dosing that could lead to more rapid and uniform healing across the treated segment,” said David Kandzari, M.D., Director of Interventional Cardiology and Chief Scientific Officer, Piedmont […]
Catheter Precision Launches Clinical Trial for Its VIVO™ Non-Invasive Mapping System
Study Expected to Expand to Five Hospitals and Evaluate Accuracy of the VIVO System LEDGEWOOD, N.J., Feb. 20, 2018 /PRNewswire/ — Catheter Precision, Inc.. announces the initiation of a pivotal clinical trial to assess the accuracy and safety of the VIVO™ (View Into Ventricular Onset) Non-Invasive Mapping System. The VIVO system […]
Mitralign raises $10m
Mitralign raises $10m FEBRUARY 21, 2018 BY BRAD PERRIELLO, MassDevice Mitralign, which is looking to commercialize its namesake mitral valve treatment and pursue regulatory nods for its Trialign tricuspid valve device, said it raised nearly $10 million. The $9.8 million funding, of a hoped-for $30.2 million, came from 19 unnamed investors beginning […]
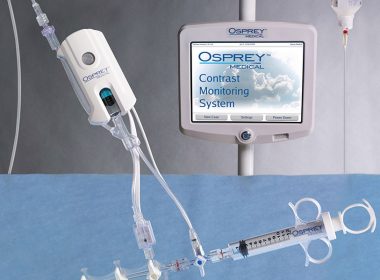
Osprey Study Chosen for “Best of Best” Presentation at Society for Cardiovascular Angiography and Interventions Conference
MINNESOTA, United States–(BUSINESS WIRE)– Osprey Medical Inc.(ASX:OSP) today announced a multi-center observational study for the DyeVert Plus™ System will feature at the Society for Cardiovascular Angiography and Interventions (SCAI) Scientific Session in April 2018. The abstract entitled: A Multi-Center Observational Study of the DyeVert Plus™ Contrast Reduction System summarizes a single-arm, multi-center observational […]
Edwards’ Self-Expanding Transcatheter Heart Valve Receives CE Mark
IRVINE, Calif., Feb. 15, 2018 /PRNewswire/ — Edwards Lifesciences(NYSE: EW), the global leader in patient-focused innovations for structural heart disease and critical care monitoring, today announced it has received CE Mark for its self-expanding CENTERA valve for severe, symptomatic aortic stenosis patients at high risk of open-heart surgery. The Edwards CENTERA […]
Bay Area’s HeartFlow Scores $240M
HeartFlow Completes Series E Financing, Securing $240 Million Proceeds Will Support Commercialization of HeartFlow® FFRct Analysis, Continued Technology Innovation and Additional Clinical Studies REDWOOD CITY, Calif.–(BUSINESS WIRE)– HeartFlow. today announced the closing of its Series E financing, securing $240 million. The company will use the proceeds from this financing to ramp up commercial […]
LivaNova Takes Out TandemLife in $250M Deal
LivaNova to Acquire TandemLife Company will add advanced cardiopulmonary temporary support solutions to its portfolio LONDON–(BUSINESS WIRE)– LivaNova PLC (NASDAQ:LIVN) (“LivaNova” or the “Company”), a market-leading medical technology company, today announced it has entered into an agreement to acquire TandemLife, a privately held company focused on advanced cardiopulmonary temporary support solutions (“TandemLife”). The […]
Claret Medical’s Sentinel Cerebral Protection System Emerging as U.S. Standard of Care for Protecting the Brain from Risk of Stroke During Transcatheter Aortic Heart Valve Replacement
Feb. 14, 2018 13:00 UTC 50 Premier U.S. Centers Adopt Sentinel Device in First Phase of Commercial Launch More Than 5,500 Patients Protected Worldwide Only Commercially Available TAVR Cerebral Protection Device in U.S., Europe and Asia Stacy Enxing Seng Joins Company’s Board of Directors SANTA ROSA, Calif.–(BUSINESS WIRE)– Since FDA […]