With current HLHS treatment, only 50-60% of infants survive to adolescenceIn ELPIS I Phase 1 clinical trial, children experienced 100% transplant-free survival up to five years of age after receiving Lomecel-B™ compared to approximate 20% mortality rate observed from historical control dataELPIS II Phase 2b clinical trial on track for completing enrollment by end of 2024Multiple nationally recognized children’s treatment centers participating in ELPIS II MIAMI, June 11, 2024 (GLOBE NEWSWIRE) — Longeveron Inc. (NASDAQ: LGVN), a clinical stage regenerative medicine biotechnology company developing cellular therapies for rare, life-threatening and chronic aging-related conditions, today announced the successful completion of an investigator meeting for ELPIS II, the on-going Phase 2b clinical trial evaluating Lomecel-B™ as a potential adjunct treatment for Hypoplastic Left Heart Syndrome (HLHS), a rare pediatric congenital heart birth defect. The investigator meeting was organized to discuss the progress to date and operational implementation of the clinical trial, inviting the principal investigator and site staff from premiere infant and children’s treatment institutions across the country. ELPIS II is on track to complete enrollment by the end of 2024. Healthcare institutions participating in the ELPIS II Phase 2 clinical trial include: Three additional nationally recognized pediatric cardiothoracic institutions participated in the investigator meeting as part of their preparation for participating as investigative sites in ELPIS II. “While we have made significant advances in diagnosing HLHS before a baby is born, we don’t yet know the cause, and current HLHS treatment requires infants to undergo a complex three-stage surgical reconstruction over the first 3-4 years of their life,” said Ram Kumar Subramanyan, division chief of Pediatric Cardiothoracic Surgery at Children’s Nebraska, and a Principal Investigator in ELPIS II. “Even with this comprehensive treatment, only 50% to 60% of infants survive to adolescence due to right ventricular failure. There is clearly an important unmet medical need to improve right ventricular function in these infants to positively impact both short- and long-term patient outcomes. Lomecel-B’s™ Phase 1 clinical data is highly encouraging and suggests it has the potential to be a new, additive treatment option for physicians and their patients dealing with HLHS. The community is very much looking forward to the results of ELPIS II.” “We believe the data generated to date in HLHS support potential for Lomecel-B™ as a regenerative medical therapy to help these infants,” said Nataliya Agafonova, MD, Chief Medical Officer of Longeveron. “In the ELPIS I Phase 1 clinical trial, participating infants experienced 100% transplant-free survival up to five years of age after receiving Lomecel-B™ compared to approximate 20% mortality rate observed from historical control data. ELPIS II is designed to further our understanding of Lomecel-B’s™ safety and efficacy in this patient population. With the collaboration of our outstanding investigative sites, we remain on track to complete enrollment of the clinical trial by the end of this year.” Longeveron’s lead investigational therapeutic candidate is Lomecel-B™, a proprietary, scalable, allogeneic, adult human bone marrow-derived medicinal signaling cell therapy. ELPIS II is enrolling 38 pediatric patients, with enrollment completion anticipated by year end 2024. The clinical trial is being conducted in collaboration with the National Heart, Lung, and Blood Institute (NHLBI) through grants from the National Institutes of Health (NIH). ELPIS II builds on the positive clinical results of ELPIS I. About Hypoplastic Left Heart Syndrome (HLHS) HLHS is a congenital birth defect in which the left ventricle (one of the pumping chambers of the heart) is either severely underdeveloped or missing. As a consequence, infants born with this condition have severely diminished systemic blood flow, which requires them to undergo a complex, three stage heart reconstruction surgery process over the course of the first 5 years of their lives. While these infants can now live into adulthood, early mortality is still extremely high in this population due to right ventricle failure, which is not meant for the increased load demanded for systemic circulation (blood circulation throughout the body). As such, there is an important unmet medical need to improve right ventricular function in these patients to improve both short-term and long-term outcomes. About Longeveron Inc. Longeveron is a clinical stage biotechnology company developing regenerative medicines to address unmet medical needs. The Company’s lead investigational product is Lomecel-B™, an allogeneic medicinal signaling cell (MSC) therapy product isolated from the bone marrow of young, healthy adult donors. Lomecel-B™ has multiple potential mechanisms of action encompassing pro-vascular, pro-regenerative, anti-inflammatory, and tissue repair and healing effects with broad potential applications across a spectrum of disease areas. Longeveron is currently pursuing three pipeline indications: hypoplastic left heart syndrome (HLHS), Alzheimer’s disease, and Aging-related Frailty. For more information, visit www.longeveron.com or follow Longeveron on LinkedIn, X, and Instagram. Forward-Looking Statements Certain statements in this press release that are not historical facts are forward-looking statements made pursuant to the safe harbor provisions of the Private Securities Litigation Reform Act of 1995, which reflect management’s current expectations, assumptions, and estimates of future operations, performance and economic conditions, and involve risks and uncertainties that could cause actual results to differ materially from those anticipated by the statements made herein. Forward-looking statements are generally identifiable by the use of forward-looking terminology such as “believe,” “expects,” “may,” “looks to,” “will,” “should,” “plan,” “intend,” “on condition,” “target,” “see,” “potential,” “estimates,” “preliminary,” or “anticipates” or the negative thereof or comparable terminology, or by discussion of strategy or goals or other future events, circumstances, or effects and include, but are not limited to, the potential for Lomecel-B™ to be a beneficial treatment for patients with HLHS. Factors that could cause actual results to differ materially from those expressed or implied in any forward-looking statements in this release include, but are not limited to, adverse global conditions, including macroeconomic uncertainty; inability to raise additional capital necessary to continue as a going concern; our history of losses and inability to achieve profitability going forward; the absence of FDA-approved allogenic, cell-based therapies for HLHS or other cardiac-related indications; ethical and other concerns surrounding the use of stem cell therapy or human tissue; our exposure to product liability claims arising from the use of our product candidates or future products in individuals, for which we may not be able to obtain adequate product liability insurance; the adequacy of our trade secret and patent position to protect our product candidates and their uses: others could compete against us more directly, which could harm our business and have a material adverse effect on our business, financial condition, and results of operations; if certain license agreements are terminated, our ability to continue clinical trials and commercially market products could be adversely affected; the inability to protect the confidentiality of our proprietary information, trade secrets, and know-how; third-party claims of intellectual property infringement may prevent or delay our product development efforts; the inability to successfully develop and commercialize our product candidates and obtain the necessary regulatory approvals; we cannot market and sell our product candidates in the U.S. or in other countries if we fail to obtain the necessary regulatory approvals; final marketing approval of our product candidates by the FDA or other regulatory authorities for commercial use may be delayed, limited, or denied, any of which could adversely affect our ability to generate operating revenues; we may not be able to secure and maintain research institutions to conduct our clinical trials; ongoing healthcare legislative and regulatory reform measures may have a material adverse effect on our business and results of operations; if we receive regulatory approval of Lomecel-B™ or any of our other product candidates, we will be subject to ongoing regulatory requirements and continued regulatory review, which may result in significant additional expense; being subject to penalties if we fail to comply with regulatory requirements or experience unanticipated problems with our therapeutic candidates; reliance on third parties to conduct certain aspects of our preclinical studies and clinical trials; interim, “topline” and preliminary data from our clinical trials that we announce or publish from time to time may change as more data become available and are subject to audit and verification procedures that could result in material changes in the final data; provisions in our certificate of incorporation and bylaws and Delaware law might discourage, delay or prevent a change in control of our company or changes in our management and, therefore, depress the market price of our Class A common stock; we have never commercialized a product candidate before and may lack the necessary expertise, personnel and resources to successfully commercialize any products on our own or together with suitable collaborators; and in order to successfully implement our plans and strategies, we will need to grow our organization, and we may experience difficulties in managing this growth. Further information relating to factors that may impact the Company’s results and forward-looking statements are disclosed in the Company’s filings with the Securities and Exchange Commission, including Longeveron’s Annual Report on Form 10-K for the year ended December 31, 2023, filed with the Securities and Exchange Commission on February 27, 2024, as amended by the Annual Report on Form 10-K/A filed March 11, 2024, its Quarterly Reports on Form 10-Q, and its Current Reports on Form 8-K. The forward-looking statements contained in this press release are made as of the date of this press release, and the Company disclaims any intention or obligation, other than imposed by law, to update or revise any forward-looking statements, whether as a result of new information, future events, or otherwise. Investor Contact:Derek ColeInvestor Relations Advisory Solutionsderek.cole@iradvisory.com
Coronary/Structural Heart
SS Innovations’ SSi Mantra Surgical Robotic System Approved for Use in Indonesia
FORT LAUDERDALE, Fla., June 11, 2024 (GLOBE NEWSWIRE) — SS Innovations International, Inc. (the “Company” or “SS Innovations”) (OTC: SSII), a developer of innovative surgical robotic technologies dedicated to making world-class robotic surgery affordable and accessible to a global population, today announced that its SSi Mantra Surgical Robotic System has received regulatory approval from the Indonesian Ministry of Health for clinical use in the Republic of Indonesia. With more than 279 million people, Indonesia is the world’s fourth-most-populous country. However, robotic surgery has been slow to reach the island nation. Indonesia’s Bunda Hospital in Jakarta acquired its first robotic surgical system in 2012 and has only completed about 700 surgeries in the past 12 years. “With the entry of the SSi Mantra to the Indonesian market, we believe that we can be instrumental in increasing access to and decreasing costs of robotic surgery options,” said Dr. Sudhir Srivastava, SS Innovation’s Chairman and CEO. “We look forward to broadening surgical horizons for Indonesians and continuing to expand our innovative surgical services and options across the globe.” “I have been in the healthcare business for the past 30 years, and this is the first time I have seen such an impressive technology,” said Mohammad Ridwan, CEO of Neuro Medika Sejahtera, the distributor of the SSi Mantra in Indonesia. “I was very satisfied to see the quality of the SSi Mantra and the advancement offered by the SSi Maya, the Company’s mixed-reality software application. I believe that SS Innovations offers a practical and cost-effective solution to help expand robotic surgery access in Indonesia.” Last month, SS Innovations announced that it had completed 100 robotic cardiac surgery cases in just over a year, utilizing the SSi Mantra Surgical Robotic System. It is the first surgical robotic system made in India, and one of the only systems in the world designed to be cost-effective with broad-spectrum surgical applications. The SSi Mantra is clinically validated in more than 70 different types of surgical procedures. SS Innovations has commenced the regulatory approval process in the United States and the European Union and anticipates receiving FDA approval to market and CE Mark approval in 2025. About SS Innovations International, Inc. SS Innovations International, Inc. (OTC: SSII) is a developer of innovative surgical robotic technologies with a vision to make the benefits of robotic surgery affordable and accessible to a larger part of the global population. SSII’s product range includes its proprietary “SSi Mantra” surgical robotic system, and “SSi Mudra” its wide range of surgical instruments capable of supporting a variety of surgical procedures including robotic cardiac surgery. SSII’s business operations are headquartered in India and SSII has plans to expand the presence of its technologically advanced, user-friendly, and cost-effective surgical robotic solutions, globally. For more information, visit SSII’s website at ssinnovations.com or LinkedIn for updates. About SSi Mantra Supporting advanced, affordable, and accessible robotic surgery, the SSi Mantra Surgical Robotic System provides the capabilities for multi-specialty usage including cardiothoracic, head and neck, gynecology, urology, general surgery and more. With its modular arm configuration, 3D 4K vision open-console design and superior ergonomics, the system engages with the surgeon and surgical teams to improve safety and efficiency during procedures. SS Innovations has partnered with Johns Hopkins University, installing the SSi Mantra at the University’s Minimally Invasive Surgical Training and Innovation Center (MISTIC) in Baltimore, MD. This installation is actively training new surgeons and promoting the global dissemination of advancements in medical robotics. The SSi Mantra system has received Indian Medical Device regulatory approval (CDSCO) and is clinically validated in India in more than 70 different types of surgical procedures. SS Innovations has commenced the regulatory approval process in the United States and the European Union and anticipates receiving FDA approval to market and CE Mark approval in 2025. Forward-Looking StatementsThis press release may contain statements that are not historical facts and are considered forward-looking within the meaning of the Private Securities Litigation Reform Act of 1995. The words “anticipate,” “assume,” “believe,” “estimate,” “expect,” “will,” “intend,” “may,” “plan,” “project,” “should,” “could,” “seek,” “designed,” “potential,” “forecast,” “target,” “objective,” “goal,” or the negatives of such terms or other similar expressions to identify such forward-looking statements. These statements relate to future events or SS Innovations International’s future financial performance and involve known and unknown risks, uncertainties and other factors that may cause our actual results, levels of activity, performance, or achievements to be materially different from any future results, levels of activity, performance or achievements expressed or implied by these forward-looking statements. For more information:PCG AdvisoryJeff Ramson jramson@pcgadvisory.com
NewAmsterdam Pharma Announces Issuance of Composition of Matter Patent for Obicetrapib by the United States Patent and Trademark Office
— New patent extends patent term for obicetrapib until July 2043 — NAARDEN, the Netherlands and MIAMI, June 11, 2024 (GLOBE NEWSWIRE) — NewAmsterdam Pharma Company N.V. (Nasdaq: NAMS or “NewAmsterdam” or the “Company”), a late-stage, clinical biopharmaceutical company developing oral, non-statin medicines for patients at risk of cardiovascular disease (“CVD”) with elevated low-density lipoprotein cholesterol (“LDL-C”), for whom existing therapies are not sufficiently effective or well-tolerated, today announced that the United States Patent and Trademark Office (“USPTO”) has issued U.S. Patent No. 12,006,305, titled “Salts of Obicetrapib and Processes for their Manufacture and Intermediates Thereof.” The newly issued patent contains claims covering amorphous obicetrapib hemicalcium, the solid form that will be used in the Company’s products and will be listed in the FDA’s “Orange Book” as a drug substance patent, if any such products are approved. The issuance of this composition of matter patent provides intellectual property (“IP”) protection for obicetrapib until July 2043. The USPTO has now issued or allowed a total of nine patents covering obicetrapib and its uses. “We are excited to announce that obicetrapib has been granted our third-generation composition of matter patent by the USPTO, which extends obicetrapib’s IP protection and exclusivity until July 2043, more than 9 years beyond our previous second-generation patent expiration in 2034,” said Michael Davidson, M.D., Chief Executive Officer of NewAmsterdam. “There continues to be a need for a safe, efficacious, oral, and potent LDL-lowering therapy. Securing robust patent protection for obicetrapib enables us both to continue the execution of multiple ongoing pivotal Phase 3 trials, as well as reap the potential benefits of obicetrapib’s scientific innovation years beyond potential FDA approval.” About Obicetrapib Obicetrapib is a novel, oral, low-dose CETP inhibitor that NewAmsterdam is developing to overcome the limitations of current LDL-lowering treatments. In each of the Company’s Phase 2 trials, ROSE2, TULIP, ROSE, and OCEAN, evaluating obicetrapib as monotherapy or combination therapy, the Company observed statistically significant LDL-lowering combined with a side effect profile similar to that of placebo. The Company is conducting two Phase 3 pivotal trials, BROADWAY and BROOKLYN, to evaluate obicetrapib as a monotherapy used as an adjunct to maximally tolerated lipid-lowering therapies to provide additional LDL-lowering for CVD patients and TANDEM, to evaluate obicetrapib and ezetimibe as a fixed-dose combination. The Company began enrolling patients in BROADWAY in January 2022, in BROOKLYN in July 2022, and in TANDEM in March 2024; completing enrollment of BROOKLYN in April 2023 and BROADWAY in July 2023. The Company also commenced the Phase 3 PREVAIL cardiovascular outcomes trial in March 2022, which is designed to assess the potential of obicetrapib to reduce occurrences of major adverse cardiovascular events, including cardiovascular death, non-fatal myocardial infarction, non-fatal stroke and non-elective coronary revascularization. NewAmsterdam completed enrollment of PREVAIL in April 2024 and randomized over 9,500 patients. About NewAmsterdam NewAmsterdam Pharma (Nasdaq: NAMS) is a late-stage biopharmaceutical company whose mission is to improve patient care in populations with metabolic diseases where currently approved therapies have not been adequate or well tolerated. We seek to fill a significant unmet need for a safe, well-tolerated and convenient LDL-lowering therapy. In multiple phase 3 studies, NewAmsterdam is investigating obicetrapib, an oral, low-dose and once-daily CETP inhibitor, alone or as a fixed-dose combination with ezetimibe, as LDL-C lowering therapies to be used as an adjunct to statin therapy for patients at risk of CVD with elevated LDL-C, for whom existing therapies are not sufficiently effective or well tolerated. Forward-Looking Statements Certain statements included in this document that are not historical facts are forward-looking statements for purposes of the safe harbor provisions under the United States Private Securities Litigation Reform Act of 1995. Forward-looking statements generally are accompanied by words such as “believe,” “may,” “will,” “estimate,” “continue,” “anticipate,” “intend,” “expect,” “should,” “would,” “plan,” “predict,” “potential,” “seem,” “seek,” “future,” “outlook” and similar expressions that predict or indicate future events or trends or that are not statements of historical matters. These forward-looking statements include, but are not limited to, statements regarding the Company’s intellectual property and its ability to enforce, and sufficiency of, its patents, business and strategic plans, the Company’s commercial opportunity, the therapeutic and curative potential of the Company’s product candidate, the Company’s clinical trials and the timing for enrolling patients, the timing and forums for announcing data, the achievement and timing of regulatory approvals, and plans for commercialization. These statements are based on various assumptions, whether or not identified in this document, and on the current expectations of the Company’s management and are not predictions of actual performance. These forward-looking statements are provided for illustrative purposes only and are not intended to serve as and must not be relied on as a guarantee, an assurance, a prediction, or a definitive statement of fact or probability. Actual events and circumstances are difficult or impossible to predict and may differ from assumptions. Many actual events and circumstances are beyond the control of the Company. These forward-looking statements are subject to a number of risks and uncertainties, including changes in domestic and foreign business, market, financial, political, and legal conditions; risks related to the approval of the Company’s product candidate and the timing of expected regulatory and business milestones, including potential commercialization; ability to negotiate definitive contractual arrangements with potential customers; the impact of competitive product candidates; ability to obtain sufficient supply of materials; global economic and political conditions, including the Russia-Ukraine and Israel-Hamas conflict; the effects of competition on the Company’s future business; and those factors described in the Company’s public filings with the Securities Exchange Commission. Additional risks related to the Company’s business include, but are not limited to: uncertainty regarding outcomes of the Company’s ongoing clinical trials, particularly as they relate to regulatory review and potential approval for its product candidate; risks associated with the Company’s efforts to commercialize a product candidate; the Company’s ability to negotiate and enter into definitive agreements on favorable terms, if at all; the impact of competing product candidates on the Company’s business; intellectual property related claims; the Company’s ability to attract and retain qualified personnel; ability to continue to source the raw materials for its product candidate. If any of these risks materialize or the Company’s assumptions prove incorrect, actual results could differ materially from the results implied by these forward-looking statements. There may be additional risks that the Company does not presently know or that the Company currently believes are immaterial that could also cause actual results to differ from those contained in the forward-looking statements. In addition, forward-looking statements reflect the Company’s expectations, plans, or forecasts of future events and views as of the date of this document and are qualified in their entirety by reference to the cautionary statements herein. The Company anticipates that subsequent events and developments may cause the Company’s assessments to change. These forward-looking statements should not be relied upon as representing the Company’s assessment as of any date subsequent to the date of this communication. Accordingly, undue reliance should not be placed upon the forward-looking statements. Neither the Company nor any of its affiliates undertakes any obligation to update these forward-looking statements, except as may be required by law. Company ContactMatthew PhilippeP: 1-917-882-7512matthew.philippe@newamsterdampharma.com Media ContactSpectrum Science on behalf of NewAmsterdamBryan BlatsteinP: 1-917-714-2609bblatstein@spectrumscience.com Investor ContactPrecision AQ on behalf of NewAmsterdamAustin MurtaghP: 1-212-698-8696Austin.Murtagh@precisionaq.com
Positive Six-Month Results from CroíValve’s TANDEM I Study to Treat Tricuspid Regurgitation Presented at New York Valves 2024
DUBLIN–(BUSINESS WIRE)–Favorable patient outcomes from the TANDEM I first-in-human clinical trial of the CroíValve DUO™ System for the percutaneous treatment of tricuspid regurgitation (TR) were presented today by Professor Wojciech Wojakowski at the New York Valves 2024 scientific conference. The DUO System is a novel transcatheter coaptation valve that works […]
Cardio Diagnostics Holdings, Inc. Achieves SOC 2 Type I Compliance Certification for Security and Confidentiality
CHICAGO–(BUSINESS WIRE)–Cardio Diagnostics Holdings, Inc. (NASDAQ: CDIO), a leader in AI-driven precision cardiovascular medicine, today announced the successful completion of its System and Organization Controls (SOC) 2 Type I audit, achieving compliance with the leading industry standards for customer data security. This milestone underscores the Company’s unwavering commitment to the highest […]
MedsEngine and NI Medical Announce Improved Cardiovascular Outcomes Utilizing NICaS Hemodynamic Measurements with MedsEngine Clinical Decision Support Software for Hypertension
DAYTON, Ohio–(BUSINESS WIRE)–MedsEngine and NI Medical are pleased to announce they will be presenting a comprehensive analysis on their management of hypertension at the European Society of Cardiology 2024 meeting in London. The impact of combining NICaS hemodynamic data with MedsEngine clinical decision support software reveals a significant reduction in […]
Morton Plant Hospital Offers Robotic-Assisted Coronary Artery Bypass Surgery
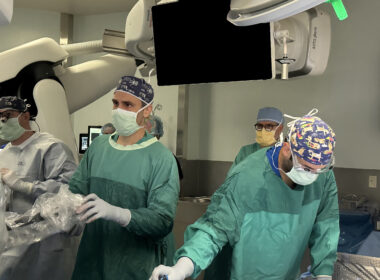
CLEARWATER, Fla., June 7, 2024 /PRNewswire/ — Morton Plant Hospital recently completed its first robotic-assisted, minimally invasive coronary artery bypass surgery (CABG), making it one of only two hospitals in the Tampa Bay area to offer robotic-assisted CABG. Morton Plant Hospital was ranked #1 in Florida for Cardiac Surgery in 2023 by Healthgrades. Cardiothoracic Surgeon Michael Kayatta, MD, and his partner, Cardiovascular Surgeon John Ofenloch, MD, performed the procedure earlier this week, with the assistance of their highly skilled surgical team.
Continue Reading
Cardiothoracic Surgeon Michael Kayatta, MD (center) works with his surgical team to prepare for Morton Plant Hospital’s first robotic-assisted coronary artery bypass surgery.
CABG surgery is needed when plaque has built up in arteries carrying blood to the heart causing the arteries to be severely narrowed. The surgeon creates a new path for blood to flow by using a healthy artery or vein from another part of the body and connects it past the blockage on the heart.
In a traditional CABG, the surgeon cuts open the chest by dividing the breastbone and spreads the ribs to expose the heart. The heart is stilled, requiring the patient to be on a heart-lung machine during the surgery. Full recovery often takes about two months. The robotic-assisted CABG only requires four small incisions between the ribs. A small camera and surgical tools are attached to the robotic arms and guided through the incisions. The heart continues to beat through the procedure, with special tools stabilizing a part of the heart.Cardiothoracic Surgeon Michael Kayatta, MD (center) works with his surgical team to prepare for Morton Plant Hospital’s first robotic-assisted coronary artery bypass surgery.The computer console, which shows images of the heart magnified up to 10 times normal vision, gives the surgeon the ability to guide the surgical instruments, attached to the robotic arms, with his fingers while watching the screen. The robotic arms that are controlled by the surgeon have greater range of motion and are more precise than human arms. A major potential benefit of a robotic-assisted CABG is a quicker recovery for the patient. The smaller incisions should also result in less scarring. “We are excited to bring this new technology to patients at Morton Plant Hospital,” said Dr. Kayatta. “Many people hear robot and think a machine will be doing the surgery. We have a highly skilled, experienced team performing each surgery. The robot is simply a tool that helps us to treat more patients and get them back to their life in weeks rather than months.” Drs. Kayatta and Ofenloch are both with BayCare Medical Group’s Cardiovascular and Thoracic Surgery clinic in Clearwater. For more information on heart and vascular services at BayCare: Heart and Vascular Services.About BayCareBayCare is a leading not-for-profit health care system that connects individuals and families to a wide range of services at 16 hospitals and hundreds of other convenient locations throughout the Tampa Bay and central Florida regions. The system is West Central Florida’s largest provider of behavioral health and pediatric services and its provider group, BayCare Medical Group, is one of the largest in the region. BayCare’s diverse network of ambulatory services includes laboratories, imaging, surgical centers, BayCare Urgent Care locations, wellness centers and one of Florida’s largest home care agencies, BayCare HomeCare. BayCare’s mission is to improve the health of all it serves through community-owned, health care services that set the standard for high-quality, compassionate care. For more information visit BayCare.org.SOURCE Morton Plant Hospital
BioCardia Announces United States Patent Issuance on Intramyocardial Delivery of Cell Aggregates to the Heart Including Minimally Invasive Radial Artery Approach
SUNNYVALE, Calif., June 07, 2024 (GLOBE NEWSWIRE) — BioCardia®, Inc. [Nasdaq: BCDA], a company focused on cellular and cell-derived therapeutics for the treatment of cardiovascular and pulmonary diseases, today announces that the Unites States Patent Office has granted Patent No: 11,986,611 titled “Radial and Transendocardial Delivery Catheter,” with a patent term that will expire in 2036. The present invention relates to medical methods and systems suitable for substance delivery to the heart via a radial artery and for the intracardiac delivery of cellular aggregates and other agglomerated materials. Radial artery delivery is a means by which cardiac catheters are advanced through a blood vessel in a patient’s wrist to treat the heart. This approach has significant advantages for patients, enabling them to leave the hospital soon after the procedure with a band aid on their wrist and their arm in a simple sling, allowing them to immediately return to their active lives. In addition, a radial approach permits hospitals and other care centers to greatly reduce costs by eliminating the need for an overnight stay by the patient. For these reasons, trans-radial access is becoming the default approach in many cardiac centers worldwide. Enabling radial access has enormous potential advantages for biotherapeutic delivery to the heart. BioCardia’s Helix system, used in our ongoing clinical trials, is the only known system with patented designs that can enable radial transendocardial biotherapeutic delivery. The issued patent claims further protect this approach and add value to both BioCardia’s therapeutic programs and those of our biotherapeutic delivery partners. Therapeutic cell aggregates have the potential advantage over single cell suspensions by enhancing retention in the heart to maximize therapeutic benefit. However, the delivery of cell aggregates carries greater risks of potentially life-threatening strokes should they leak into the ventricular chamber. As 20% of the blood in the heart chamber goes to the brain, 20% of any cell or cell aggregates released in the heart are expected to obstruct the first cerebral artery that is too small to allow the cell or cell aggregate to pass. The larger the cell aggregate, the larger the vessel they are capable of obstructing, with greater risk of a significant life-threatening stroke. BioCardia’s delivery systems are designed to prevent leakage of therapeutic agents into the ventricular chamber by providing stable and safe engagement to the heart tissue during the delivery process. The issued patent claims further protect this design and add value to both BioCardia’s therapeutic programs and those of our biotherapeutic delivery partners. “Our minimally invasive biotherapeutic delivery platforms enable the successful development of cell and gene-based therapies for the heart,” said Dr. Peter Altman, BioCardia CEO. “The Helix platform underlies BioCardia’s cell therapy clinical programs and this recent patent issuance provides additional protection to our technology and product offerings in the United States for at least another dozen years. This is just one of many patent applications we are advancing to protect our value creation for the benefit of shareholders, which in turn enables these advances to be supported for expected benefit to millions of patients.” About BioCardia® BioCardia, Inc., headquartered in Sunnyvale, California, is developing cellular and cell-derived therapeutics for the treatment of cardiovascular and pulmonary diseases. CardiAMP™ autologous and CardiALLO allogeneic cell therapies are the Company’s biotherapeutic platforms in three clinical stage product candidates in development. BioCardia also partners with other biotherapeutic companies to provide its delivery systems and development support to their programs. For more information visit: www.BioCardia.com. Forward Looking Statements: This press release contains forward-looking statements that are subject to many risks and uncertainties. Forward-looking statements include, among other things, references to the Company’s investigational product candidates, the benefits and risks of cell aggregates, the benefits of radial artery access, the level and duration of patent protection that the Company’s patents provide to its product candidates, and the expected safety of BioCardia’s delivery device designs. These forward-looking statements are made as of the date of this press release, and BioCardia assumes no obligation to update the forward-looking statements. We may use terms such as “believes,” “estimates,” “anticipates,” “expects,” “plans,” “intends,” “may,” “could,” “might,” “will,” “should,” “approximately” or other words that convey the uncertainty of future events or outcomes to identify these forward-looking statements. Although we believe that we have a reasonable basis for each forward-looking statement contained herein, we caution you that forward-looking statements are not guarantees of future performance and that our actual results may differ materially from the forward-looking statements contained in this press release. As a result of these factors, we cannot assure you that the forward-looking statements in this press release will prove to be accurate. Additional factors that could materially affect actual results can be found in BioCardia’s Form 10-K filed with the Securities and Exchange Commission on March 27, 2024, under the caption titled “Risk Factors” And in its subsequently filed Quarterly Reports on Form 10-Q. BioCardia expressly disclaims any intent or obligation to update these forward-looking statements, except as required by law.
University Hospitals Now Performing Robotic Heart Bypass Surgeries
CLEVELAND – A new, robotic approach to heart bypass surgery is now being offered to patients at University Hospitals (UH) with great success. Two UH Harrington Heart & Vascular Institute providers, Dr. Kelsey Gray and Dr. Pablo Ruda Vega, are currently the only surgeons in Ohio performing this specific type […]
Hypertrophic Cardiomyopathy 101
What every student-athlete should knowMISSION, Kan., June 6, 2024 /PRNewswire/ — (Family Features) You may find it difficult to wrap your mind around the idea of an energetic student-athlete with a cardiac diagnosis. Heart conditions may be more often associated with older individuals, but you might be surprised to learn hypertrophic cardiomyopathy is the most common condition responsible for sudden cardiac death in young athletes. In fact, it’s the cause of 40% of sudden cardiac death cases.
Continue Reading
Photo courtesy of Shutterstock
Photo courtesy of Shutterstock
It’s estimated 1 in every 500 adults living in the United States has hypertrophic cardiomyopathy, according to the American Heart Association, but a significant percentage are undiagnosed. More than 80% of individuals who experience this condition show no signs or symptoms before sudden cardiac death. While sudden cardiac death is rare, it can occur during exercise or in its aftermath. That’s why it’s important for student-athletes and their loved ones to learn more about this condition and talk to a doctor about their risk.
With proper knowledge and the support of a skilled care team, it’s possible to manage hypertrophic cardiomyopathy with heart-healthy actions to prevent complications or worsening cardiovascular conditions like atrial fibrillation (a quivering or irregular heartbeat), stroke or heart failure. Hypertrophic cardiomyopathy awareness and education for athletes by the American Heart Association is made possible in part by a grant from the Bristol Myers Squibb Foundation.
What is hypertrophic cardiomyopathy?Hypertrophic cardiomyopathy is the most common form of inherited heart disease and can affect people of any age. It’s defined by thickening and stiffening of the walls of the heart. The heart’s chambers cannot fill up or pump blood out adequately, so the heart is unable to function normally.There are different types of this condition. Most people have a form of the disease in which the wall that separates the two bottom chambers of the heart (the septum) becomes enlarged and restricts blood flow out of the heart (obstructive hypertrophic cardiomyopathy).However, sometimes hypertrophic cardiomyopathy occurs without significant blocking of blood flow (nonobstructive hypertrophic cardiomyopathy). The heart’s main pumping chamber is still thickened and may become increasingly stiff, reducing the amount of blood taken in then pumped out to the body with each heartbeat.What are possible symptoms?Symptoms can include:
shortness of breath
chest pain
heart palpitations
fatigue
The severity of symptoms can vary, but if you experience them or if you have a family history of hypertrophic cardiomyopathy or sudden cardiac death, it may be a good idea to speak to your doctor about whether you have this condition.For some people, symptoms can get worse and new symptoms can appear over time, resulting in people dealing with harsher effects and a diminished ability to do the activities they love. This decrease in functions can be one of the most challenging aspects of the disease. Keeping your health care team aware of any new or changing symptoms allows them to work with you to develop a plan to manage these symptoms and reduce their impact.How is hypertrophic cardiomyopathy diagnosed?Medical history, family history, a physical exam and diagnostic test results all factor into a diagnosis. A common diagnostic test is an echocardiogram that assesses the thickness of the heart muscle and observes blood flow from the heart.If anyone in your family has been diagnosed with hypertrophic cardiomyopathy, other heart diseases or has been told they had thick heart walls, you should share that information with your doctor and discuss the need for genetic testing. Because this condition is hereditary, first-degree relatives, which include siblings and parents, should be checked.Learn more at heart.org/HCMStudentAthlete.Photos courtesy of ShutterstockMichael French[email protected]1-888-824-3337editors.familyfeatures.comAbout Family Features Editorial SyndicateA leading source for high-quality food, lifestyle and home and garden content, Family Features provides readers with topically and seasonally relevant tips, takeaways, information, recipes, videos, infographics and more. Find additional articles and information at Culinary.net and eLivingToday.com.SOURCE Family Features Editorial Syndicate