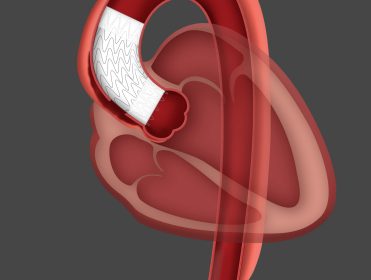
FLAGSTAFF, Ariz.–(BUSINESS WIRE)–W. L. Gore & Associates, Inc. (Gore) today announced the first implant in conjunction with the Gore ARISE Study of the GORE® Ascending Stent Graft, an investigational device and the only endovascular stent graft specifically designed to treat Type A dissections of the ascending aorta. The successful procedure took place […]
Peripheral/Endo
GORE® Molding & Occlusion Balloon for Endovascular Aortic Repair Receives Approval in the United States, Japan, and Europe
FLAGSTAFF, Ariz.–(BUSINESS WIRE)–W. L. Gore & Associates, Inc. (Gore) announced FDA 510(k) clearance, approval from the Japanese Ministry of Health, Labour, and Welfare, and receipt of CE Mark for the innovative GORE® Molding & Occlusion Balloon, a compliant polyurethane balloon catheter designed in close collaboration with clinicians to assist in the expansion […]
Vascular Graft Solutions Reports the First Clinical Use of Its FRAME External Support Technology in Aneurysm Repair of High Flow Arteriovenous Fistulas
TEL AVIV, Israel, Aug. 20, 2018 /PRNewswire/ — Vascular Graft Solutions reports the first clinical use of its FRAME external support technology for aneurysm repair in high flow arteriovenous (AV) fistulas. The first 5 cases were performed by Dr. Vladimir Matoussevitch from Cologne University Hospital in Germany. “Autogenous AV fistula access for hemodialysis is a […]
NuCryo Vascular Announces Commercialization Agreement with Lokai Medical for Innovative Treatment for Peripheral Arterial Disease
SAN JOSE, Calif.–(BUSINESS WIRE)–NuCryo Vascular today announced that the company has signed a commercialization agreement with Lokai Medical, a specialty distributor of coronary/peripheral and interventional devices, to distribute the PolarCath™ Balloon Dilatation System in the United States. Balloon Cryoplasty® has been shown in clinical studies and in everyday procedures to be […]
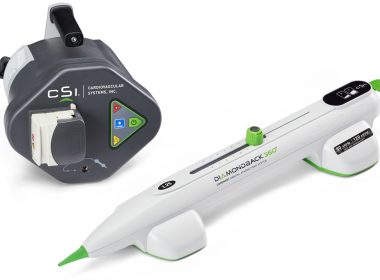
Cardiovascular Systems Presents Liberty 360° Two-Year Outcomes at 2018 Amputation Prevention Symposium
ST. PAUL, Minn.–(BUSINESS WIRE)–Cardiovascular Systems, Inc. (CSI®) (NASDAQ: CSII), a medical device company developing and commercializing innovative interventional treatment systems for patients with peripheral and coronary artery disease, presented two-year outcomes from its LIBERTY 360° study in a late-breaking presentation at the 2018 Amputation Prevention Symposium (AMP) in Chicago. The […]
Cook Medical Introduces a New Length of Zilver® PTX®
BLOOMINGTON, Ind.–(BUSINESS WIRE)–Cook Medical has introduced the 140 mm-length Zilver® PTX® Drug-Eluting Peripheral Stent in both 6 and 7 mm diameters in the U.S. The longer length comes after an expanded indication approval by FDA to treat total lesion lengths up to 300 mm per patient. In addition, the product […]
Boston Scientific Announces Agreement to Acquire VENITI, Inc.
MARLBOROUGH, Mass., Aug. 8, 2018 /PRNewswire/ — Boston Scientific Corporation (NYSE: BSX) today announced it has signed an agreement to acquire VENITI, Inc., a privately-held company in Fremont, California which has developed and commercialized the VICI VENOUS STENT® System for treating venous obstructive disease. Boston Scientific has been an investor in VENITI since 2016 and currently […]
NexGen Medical Systems, Inc. Launches XCOIL® Large Vessel (18mm) Thrombectomy System
MINNEAPOLIS, Minn., August 6th,2018/ /– NexGen Medical Systems, Inc., a Minneapolis medical device company, announced today the successful completion of the first human use of their XCOIL® Large Vessel (18mm) Thrombectomy System for the treatment of deep vein thrombosis (DVT). The new device, an expansion of the successful XCOIL® 12mm […]
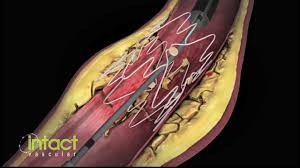
Symposium to Explore the Impact of Vascular Dissections on Critical Limb Ischemia Patients at AMP 2018
WAYNE, Pa.–(BUSINESS WIRE)–Intact Vascular, Inc., a developer of medical devices for minimally invasive peripheral vascular procedures, today announced key presentations that will be featured during a CME symposium at the Amputation Prevention (AMP) Conference in Chicago on August 8. The symposium, titled “Why Dissections Matter: A case-based look at below-the-knee […]
Corindus to Showcase CorPath GRX® System for Peripheral Vascular Interventions at the 2018 Amputation Prevention Symposium
WALTHAM, Mass.–(BUSINESS WIRE)–Corindus Vascular Robotics, Inc. [NYSE American: CVRS], a leading developer of precision vascular robotics, announced today that the CorPath GRX System will be showcased for peripheral applications at the 2018 Amputation Prevention Symposium in Chicago, IL. Jihad Mustapha, M.D., Interventional Cardiologist and AMP course director, will moderate an […]