Phase 3 DiSCOVER clinical trial initiation expected by end of Q2 2022 NEWTON, Mass., May 09, 2022 (GLOBE NEWSWIRE) — Acer Therapeutics Inc. (Nasdaq: ACER), a pharmaceutical company focused on the acquisition, development and commercialization of therapies for serious rare and life-threatening diseases with significant unmet medical needs, today announced […]
Peripheral/Endo
Verve Therapeutics Reports Additional VERVE-101 and GalNAc-Lipid Nanoparticle Delivery Data in Non-Human Primates at TIDES USA 2022
VERVE-101 Preclinical Data Package Supports Company’s Global Development Strategy and Regulatory Submissions for Clinical Trials in Patients with HeFH Single Intravenous Infusion of VERVE-101 Clinical Formulation Led to Mean 68% Reduction in Plasma LDL-C in NHPs Out to One-Year with Good Tolerability; VERVE-101 Precursor Formulation Led to Durability of LDL-C […]
Access Vascular Receives FDA Clearance for HydroPICC® Biomaterial-Based Dual-Lumen Catheter
Single- and dual-lumen biomaterial catheters allow hospitals to reduce thrombosis and vascular access complications for majority of PICC applications BILLERICA, Mass., May 05, 2022 (GLOBE NEWSWIRE) — Access Vascular, Inc. (AVI) today announced that it has received U.S. Food and Drug Administration (FDA) 510(k) clearance for its HydroPICC® Dual-Lumen catheter. Designed […]
Silk Road Medical Announces FDA Approval of Expanded Indications for the ENROUTE® Transcarotid Stent System
SUNNYVALE, Calif., May 02, 2022 (GLOBE NEWSWIRE) — Silk Road Medical, Inc. (Nasdaq: SILK), a company focused on reducing the risk of stroke and its devastating impact, today announced that that the U.S. Food and Drug Administration (FDA) approved expanded indications for the ENROUTE stent to include patients at standard […]
Biomerics Announces Image Guided Intervention (IGI) Division
SALT LAKE CITY, May 2, 2022 /PRNewswire/ — Biomerics, a leading vertically integrated contract manufacturer for the attractive interventional device market, announced the creation of a new division, Biomerics Image Guided Intervention (IGI). Biomerics’ IGI Division, headquartered in Salt Lake City, provides advanced image generation and processing technologies to enable real-time minimally invasive […]
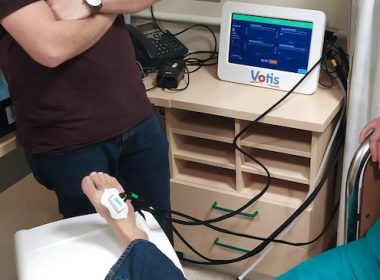
VOTIS SUBDERMAL IMAGING TECHNOLOGIES, LTD. ANNOUNCES ENROLLMENT OF FIRST HUMAN SUBJECT IN PILOT FEASIBILITY STUDY OF PEDCHECK™ AT HADASSAH MEDICAL CENTER IN JERUSALEM
JERUSALEM, May 2, 2022 /PRNewswire/ — VOTIS Subdermal Imaging Technologies Ltd. announced today that it has enrolled the first patient in the feasibility study of its PedCheckTM device at Hadassah Medical Center in Jerusalem, Israel. The pilot study, entitled “The Use of Hemodynamic Occlusive Vascular Response (HOVR™) technology to Screen for Peripheral Arterial Disease (PAD)”, is intended to […]
Boston Scientific Receives FDA Clearance for the EMBOLD™ Fibered Detachable Coil
MARLBOROUGH, Mass., April 28, 2022 – Boston Scientific Corporation (NYSE: BSX) has received U.S. Food and Drug Administration (FDA) 510(k) clearance for the EMBOLD™ Fibered Detachable Coil, which is indicated to obstruct or reduce the rate of blood flow in the peripheral vasculature. The first procedure using the device was performed this week […]
Koya – Medical Device Innovations Company Launches Active Compression System for Lymphedema and Venous Disease – UCSF Rosenman Institute
Oakland, California, 2022-Apr-28 — /EPR Network/ — Koya Medical announced today that it commercially launched its Dayspring active compression system in the U.S. Oakland, California-based Koya designed the wearable Dayspring compression system to treat lymphedema and venous diseases in the lower extremities. It features a soft, breathable mesh garment with Koya’s proprietary Flexframe […]
Pounce™ Thrombectomy System First-in-Human Data Show 100 Percent Technical Success in Early Cases
Thrombolytics in target lesion were not used for 19 of 20 patients treated with next-generation mechanical arterial thrombectomy device EDEN PRAIRIE, Minn.–(BUSINESS WIRE)–Surmodics, Inc. (NASDAQ : SRDX), a leading provider of medical device and in vitro diagnostic technologies to the health care industry, announced today that its Pounce™ Thrombectomy System […]
Access Vascular Inc. Wins 2022 Edison Award for Innovation
Access Vascular’s biomaterial catheters recognized as Best New Product in Science & Medical category for their ability to dramatically improve vascular access procedures and outcomes BILLERICA, Mass., April 26, 2022 (GLOBE NEWSWIRE) — Access Vascular, Inc. (AVI) today announced its biomaterial-based hydrophilic catheters received a Gold Award as the 2022 Edison […]