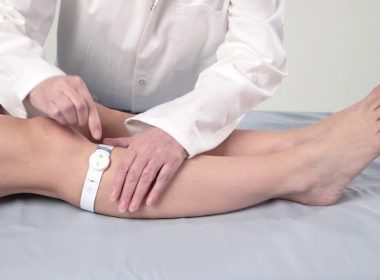
With this move, government hospitals across India can now access the geko™ device to prevent life threatening blood clots, address complications related to swelling following orthopaedic surgery and the healing of chronic wounds (leg ulcers). DARESBURY, England, April 6, 2022 /PRNewswire/ — Sky Medical Technology Ltd (Sky), a UK-based medical device manufacturer and parent company […]
Peripheral/Endo
Ra Medical Systems Granted U.S. Patent for Laser Ablation Catheters with Expanded Ablation Area
CARLSBAD, Calif.–(BUSINESS WIRE)–Ra Medical Systems, Inc. (NYSE American: RMED), a medical device company focusing on developing its excimer laser system to treat vascular diseases, announces the United States Patent and Trademark Office has granted the company a patent for liquid-filled laser ablation catheters with expanded distal optical windows. U.S. patent […]
Data from New VOYAGER PAD Analyses at ACC.22 Reinforce Benefit of XARELTO® (rivaroxaban) Plus Aspirin in Patients with Peripheral Artery Disease (PAD) and Various Co-Morbid Conditions
An analysis showed benefit of XARELTO® plus aspirin in reducing thrombotic hospitalizations for PAD patients with and without chronic kidney disease (CKD) A separate analysis demonstrated PAD patients who received XARELTO® plus aspirin in addition to statin therapy had the lowest risk of the composite of major adverse cardiovascular events (MACE) or major adverse […]
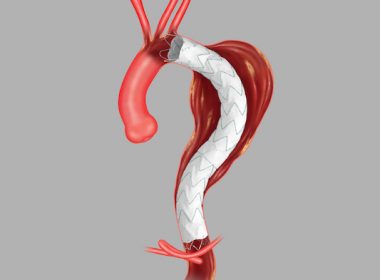
Cook Medical receives FDA Breakthrough Device Designation for Zenith® Thoraco+ Endovascular System
Bloomington, Ind. — Cook Medical’s Zenith® Thoraco+ Endovascular System (Thoraco+) has received Breakthrough Device Designation from the US Food and Drug Administration (FDA). This designation is granted to devices that have the potential to provide more effective treatment or diagnosis for life-threatening or irreversibly debilitating diseases or conditions. While the product is […]
New Janssen Initiative Aims to Advance Equitable Care and Address Hidden Threat of Amputation Related to Peripheral Artery Disease (PAD)
Through Save Legs. Change Lives.™, Janssen seeks to elevate PAD research, collaboration, education and screening for communities placed at increased risk of cardiovascular disease TITUSVILLE, N.J., March 31, 2022 /PRNewswire/ — The Janssen Pharmaceutical Companies of Johnson & Johnson today announced the launch of Save Legs. Change Lives.™ Spot Peripheral Artery Disease Now, a multi-year […]
Shockwave Medical Announces Global Launch of New Peripheral Intravascular Lithotripsy Catheter
Shockwave M5+ IVL Catheter Broadens Options for Treatment of Peripheral Arteries SANTA CLARA, Calif., March 31, 2022 (GLOBE NEWSWIRE) — Shockwave Medical, Inc. (NASDAQ: SWAV), a pioneer in the development of Intravascular Lithotripsy (IVL) to treat severely calcified cardiovascular disease, today announced the global commercial availability of the Shockwave M5+ peripheral […]
Nipro introduces second-gen Cronus® HP PTA balloon catheter to U.S. market – a unique bridge of renal and vascular divisions.
BRIDGEWATER, N.J., March 30, 2022 /PRNewswire/ — Nipro Medical Corporation (Nipro), a leading manufacturer and supplier of renal, vascular, and medical-surgical products, welcomes a second-gen Cronus® HP PTA Balloon Catheter to the U.S. Cronus® HP is a high pressure (HP) percutaneous transluminal angioplasty (PTA) balloon catheter indicated for use in popliteal, femoral, iliac, and renal […]
Hyperflex® Balloon Catheter by Endovastec™ Receives Marketing Approval in Japan
SHANGHAI, March 28, 2022 /PRNewswire/ — Shanghai MicroPort Endovascular MedTech (Group) Co., Ltd. (Endovastec™) recently announced that it has received registration approval from Japan Pharmaceuticals and Medical Devices Agency (PMDA) for its independently-developed Hyperflex® Balloon Catheter (Hyperflex®) as the company’s first product approved for marketing in Japan. Hyperflex® obtained CE Mark in 2016 […]
First-in-Man Use of the Tractus™ Crossing Support Catheter with its Jigsaw Technology™ Shaft
EATONTOWN, N.J.–(BUSINESS WIRE)–Tractus Vascular, LLC today announced the first-in-man use of the TractusTM Crossing Support Catheter (TractusTM CSC). The TractusTM CSC represents a highly novel approach to treating vascular disease. Chronic total occlusion (CTO) remains one of the most challenging pathologies encountered by surgeons and interventionalists in performing endovascular interventions. Drs. John H. Rundback […]
Einstein Researchers Find New Strategy for Preventing Clogged Arteries
BRONX, N.Y., March 28, 2022 /PRNewswire/ — Revving up a process that slows down as we age may protect against atherosclerosis, a major cause of heart attacks and strokes. In findings publishing online this week in Proceedings of the National Academy of Sciences (PNAS), scientists at Albert Einstein College of Medicine led by Ana Maria Cuervo, M.D., Ph.D., successfully […]