New Chinese Distributor Will Also Invest in At-The-Market Private Placement TEL AVIV, Israel, Feb. 03, 2021 (GLOBE NEWSWIRE) — InspireMD, Inc. (NYSE American: NSPR), developer of the CGuard™ Embolic Prevention System (EPS) for the prevention of the stroke caused by carotid artery disease, today announced a transaction intended to achieve […]
Peripheral/Endo
Cardiovascular Systems, Inc. Announces Partnership With Chansu Vascular Techologies, LLC to Develop New Drug-Coated Balloon Technology
ST. PAUL, Minn.–(BUSINESS WIRE)–Cardiovascular Systems, Inc. (CSI®) (NASDAQ: CSII) announced today that it has partnered with Chansu Vascular Technologies, LLC (CVT) to develop novel peripheral and coronary everolimus drug-coated balloons (DCBs). This partnership brings together CSI’s leadership in the treatment of calcified arterial lesions with Dr. Philippe Marco, a pioneer […]
First-of-its-Kind Randomized Clinical Trial Published in “The Lancet Infectious Diseases” Outlines Potential New Standard-Setting Best Practices for Peripheral IV Catheters
– There is robust evidence that the BD vascular care solution and skin antiseptic for peripheral intravenous catheters used in the CLEAN3 randomized clinical trial can help improve patient outcomes – Compared with standard approaches, the BD vascular care solution reduced the relative risk of catheter failure by 27%, resulting […]
Cagent Vascular Announces PRELUDE-BTK Study Results Presented at LINC Symposium: Confirms Serranator Device Novel Mechanism of Action
WAYNE, Pa.–(BUSINESS WIRE)–Cagent Vascular, a developer of serration technology for vessel dilatation in endovascular interventions, announced the results of its PRELUDE-BTK Study at this year’s LINC Symposium on January 29th. The PRELUDE-BTK Study, was a prospective, single-arm, multi-center feasibility study to show the safety and efficacy of Serration Angioplasty. The […]
12-Month Below-the-Knee Data with MedAlliance’s SELUTION SLR™ Presented as Late Breaking Trial at LINC
NYON, Switzerland, Jan. 28, 2021 /PRNewswire/ — 12-month results from the PRESTIGE* Below-the-Knee (BTK) study have been presented as a Late Breaking Trial at LINC 2021. The objective of this clinical investigation has been to evaluate safety and performance outcomes of SELUTION SLR™, MedAlliance’s novel sirolimus-eluting balloon, for the treatment of long tibial occlusive […]
Results of the FLEX Vessel Prep ™ system were presented at the Leipzig Interventional Course (LINC) 2021
Three studies show that the FLEX Vessel Prep System safely and effectively modifies obstructive plaque to facilitate definitive therapies MINNEAPOLIS , Jan. 28, 2021 / PRNewswire / – VentureMed Group, Inc. (VentureMed), a privately held innovative medical technology company, today announced that the results of three studies examining the use of the FLEX Vessel Prep […]
Artio Medical Announces Successful First Human Use of the Amplifi™ Vein Dilation System
First-of-its-kind medical device aims to improve vascular access for hemodialysis patients PRAIRIE VILLAGE, Kan., Jan. 27, 2021 /PRNewswire/ — Artio Medical, Inc., a medical device company developing innovative products for the peripheral vascular, neurovascular, and structural heart markets, announced today that it has successfully completed the first human use of its Amplifi™ Vein Dilation […]
FLEX Vessel Prep™ System Data Presented During Leipzig Interventional Course (LINC) 2021
Trio of studies demonstrate that FLEX Vessel Prep System safely and effectively modifies obstructive plaque to facilitate delivery of definitive therapies MINNEAPOLIS, Jan. 27, 2021 /PRNewswire/ — VentureMed Group, Inc. (VentureMed), a privately-held medical device innovator, announced today that data from three studies evaluating the use of its FLEX Vessel Prep (VP) System were […]
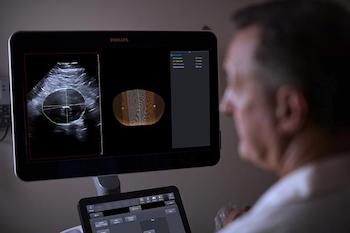
Philips integrates 3D ultrasound with innovative software for breakthrough in surveillance of abdominal aortic aneurysms
Philips Abdominal Aortic Aneurysm (AAA) Model helps increase diagnostic confidence and improved patient experience compared to current standard of care Amsterdam, the Netherlands – Royal Philips (NYSE: PHG, AEX: PHIA), a global leader in health technology, has introduced the Philips Abdominal Aortic Aneurysm (AAA) Model, providing physicians a more patient-friendly solution compared to […]
SoundBite Medical Solutions Announces First Use of its Novel 0.014” Active Wire to Successfully Treat Calcified Below-The-Knee CTOs
MONTREAL–(BUSINESS WIRE)–SoundBite Medical Solutions Inc. (SBMS) announced today the first use of its novel Active Wire 0.014” platform in the successful treatment of patients suffering from critical limb ischemia (CLI) with heavily calcified below-the-knee (BTK) chronic total occlusions (CTO). The procedures were performed by Professor Marianne Brodmann, Head of the […]