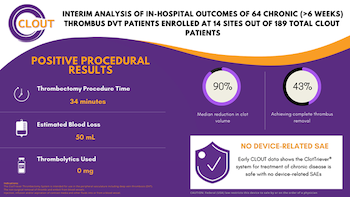
IRVINE, Calif., Jan. 25, 2021 (GLOBE NEWSWIRE) — Inari Medical, Inc. (NASDAQ: NARI) (“Inari”) a commercial-stage medical device company focused on developing products to treat and transform the lives of patients suffering from venous diseases, today announced strongly positive interim results of the first 64 chronic deep vein thrombosis (“DVT”) […]
Peripheral/Endo
PEDRA™ Technology Receives FDA Breakthrough Device Designation for its PEDRA™ Xauron™ Real-Time Tissue Perfusion System
Novel perfusion monitor achieves FDA Breakthrough Device Designation for real-time, periprocedural monitoring of tissue perfusion in patients with critical limb threatening ischemia SINGAPORE, Jan 25, 2021 /PRNewswire/ — PEDRA™ Technology, a privately-held company, announced today that the U.S Food and Drug Administration (FDA) has granted the company a Breakthrough Device Designation for the […]
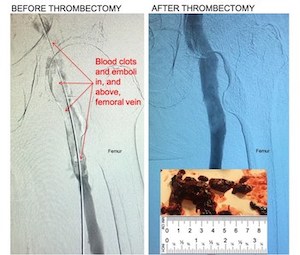
Kishor Vora, M.D. Owensboro Heart & Vascular, Removes Large Blood Clots in 1st Global Use of New Device
11F mechanical thrombectomy system removes large blood clots in deep vein thrombosis (DVT) HALLANDALE BEACH, Flo., Jan. 22, 2021 /PRNewswire/ — Control Medical Technology announced the FDA cleared Control 11F Mechanical Thrombectomy system was used to remove large blood clots from patients with deep vein thrombosis (DVT). “Control removed large blood […]
JACC Study: PEN Indigo Aspiration System Meets Safety/Efficacy Endpoints
A study published online first in the JACC: Cardiovascular Interventions found that Penumbra, Inc.’s Indigo Aspiration System met its predefined safety and efficacy endpoints for the treatment of pulmonary embolism (PE) in the EXTRACT-PE study. Specific results include a significant mean reduction in right ventricular (RV)/left ventricular (LV) ratio of […]
Teleflex continues to deliver COMPLETE confidence with new Arrow® ErgoPack® Complete MAC, PSI Systems
Teleflex expands value of ErgoPack® Complete enhancements to critical vascular access products WAYNE, Pa., Jan. 19, 2021 (GLOBE NEWSWIRE) — Teleflex Incorporated (NYSE: TFX) continues to deliver COMPLETE confidence with the release of the Arrow® ErgoPack® Complete Multi-Lumen Access Catheter (MAC) System and the Arrow® ErgoPack® Complete Percutaneous Sheath Introducer (PSI) System. The release […]
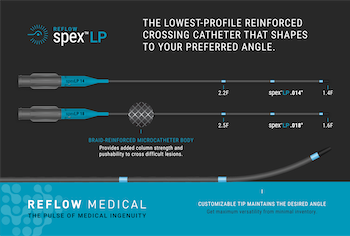
Reflow Medical Introduces the Spex™ LP, the Lowest Profile Shapeable Reinforced Support Catheter
SAN CLEMENTE, Calif.–(BUSINESS WIRE)–Reflow Medical, Inc., a California-based medical device company, introduces the Reflow™ Spex™ LP (Low Profile) 0.014 and 0.018-inch reinforced support catheters. The new Spex LP is engineered to provide the lowest profile tip for accessing and crossing the tightest and most complex lesions with a supportive system. It also […]
Microbot Medical Announces Positive Results of Additional Animal Feasibility Study with LIBERTYTM Robotic System
Results Validate Findings of Initial Study; End Points Achieved with No Intraoperative Adverse Events Management Presenting at Needham Growth Conference Today at 2:00pm ET HINGHAM, Mass., Jan. 14, 2021 (GLOBE NEWSWIRE) — The continued progression of Microbot Medical Inc.’s (Nasdaq: MBOT) LIBERTY™ Robotic System was further demonstrated as the Company announced […]
Marizyme Announces Approval for DuraGraft® in India
Latest Approval Joins a Growing List of Countries for Commercial Sales in 2021 JUPITER, Fla., Jan. 14, 2021 /PRNewswire/ — Marizyme, Inc. (OTCQB:MRZM), a publicly traded global biotechnology company developing products to reduce the burden of ischemia-reperfusion injury in tissue grafting, organ transplant, and other surgical indications, announced today that its flagship product DuraGraft® is […]
GBT Continues Research on AI Detection System
SAN DIEGO, Jan. 07, 2021 (GLOBE NEWSWIRE) — GBT Technologies Inc. (OTC PINK: GTCH) (“GBT”, or the “Company”), announced that its joint venture, GBT Tokenize Corp. (“GBT/Tokenize”), is continuing with its research associated with an AI system to detect and open clogs within arteries based on its proprietary radio technology, which has an […]
VOTIS Subdermal Imaging Technologies And ii Ventures Announce Plans To Develop Preventative Medicine Devices To Screen For Peripheral Artery Disease In India
An estimated 41-54 million Indians have the disease, many undiagnosed Emphasis on impoverished rural sector, comprising 65.53% of the national population JERUSALEM and MUMBAI, India, Dec. 21, 2020 /PRNewswire/ — VOTIS Subdermal Imaging Technologies, Ltd., an Israeli corporation, and ii Ventures Private Limited (iiV), an Indian company, announced today that they have entered into a […]