MENLO PARK, Calif., March 19, 2026 /PRNewswire/ — Endovascular Engineering, Inc. (“E2”) today announced that two abstracts related to the ENGULF study and the Hēlo® Platform have been accepted as late-breaking podium presentations at the Society of Interventional Radiology (SIR) Annual…
Radiology
United Imaging Intelligence at ECR 2026: Validating, Expanding, and Applying Radiology AI at Scale
SHANGHAI, March 12, 2026 /PRNewswire/ — At ECR 2026, under the theme “Rays of Knowledge”, United Imaging Intelligence (UII) showcased a comprehensive suite of pioneering AI solutions, demonstrating how radiology expertise delivers real value when it is clinically validated,…
Dynamic Access Acquires PICC Stat in Minnesota
DALLAS, March 10, 2026 /PRNewswire/ — Dynamic Access, LLC announced today the acquisition of PICC Stat, a respected, family-owned vascular access provider based in Minneapolis and recognized for its commitment to high-quality, patient-centered care. PICC Stat built a strong reputation…
GE HealthCare announces U.S. FDA 510(k) clearance for View, a next‑generation diagnostic viewer enabling anywhere‑access to radiologists
CHICAGO–(BUSINESS WIRE)–GE HealthCare today announced that View, the powerful viewer within the Genesis™ Radiology Workspace, has received 510(k) clearance from the U.S. Food and Drug Administration (FDA). Designed as a fast, diagnostic, zero‑footprint viewer, View serves as the core anchor of the Genesis Radiology Workspace.2 This next‑generation solution aims to transform radiology workflows, unify the user experience, and empower radiologists with greater efficiency and precision. Studies
Microbot Medical® Aligns with Recently Adopted Policy of the Largest U.S.-Based Medical Association to Expand Protection for Healthcare Professionals from Ionizing Radiation
LIBERTY® Endovascular Robotic System’s Capability to Reduce Radiation Exposure Appeals to Hospitals and Healthcare ProvidersHINGHAM, Mass., March 04, 2026 (GLOBE NEWSWIRE) — Microbot Medical Inc. (Nasdaq: MBOT), developer and distributor of the innovative LIBERTY® Endovascular Robotic System, commends the American Medical Association’s (AMA) expanding efforts to protect health care professionals from ionizing radiation. In late 2025, the AMA adopted a new policy to strengthen protections for health care professionals and trainees who may face occupational radiation exposure. The policy emphasizes the importance of continued research into the health effects of cumulative exposure to ionizing radiation, the effectiveness of Personal Protection Equipment (PPE), and education and training to minimize occupational risk to physicians and to their patients. Microbot believes that this guidance underscores the growing need for solutions that enhance safety in interventional procedures, creating meaningful opportunities for technologies like LIBERTY. The LIBERTY System is remotely operated, enabling physicians and staff to perform procedures away from the radiation source. In the ACCESS-PVI Study, using LIBERTY demonstrated a 92% relative reduction in radiation exposure. Prolonged exposure to radiation often has long-term effects that can lead to a host of health issues, including cancer, cardiovascular disease, reproductive health effects, and cataracts. This risk has also been cited as contributing to the staffing shortage in the endovascular space, and especially in interventional radiology, which currently ranks number two among specialties with highest physician shortages. It disproportionately affects women, who often cite radiation exposure and the physical demands of wearing lead aprons as barriers to entering or advancing in the field. The LIBERTY System’s wireless operated capabilities allow healthcare providers to position themselves away from the radiation source and operate in a seated position, reducing reliance on heavy PPE, thereby lessening musculoskeletal strain. “Prolonged exposure to radiation and staffing shortages are rising concerns among healthcare professionals in the endovascular space,” commented Harel Gadot, CEO, President and Chairman. “We believe that these challenges strain an already overburdened health care system, impacting patient care, and widening the skills gap required to address the medical concerns. We also believe that LIBERTY can play a major role in addressing these challenges and supporting better care for both providers and their patients.” LIBERTY is the only FDA cleared, single-use, remotely operated robotic system for peripheral endovascular procedures, and it is designed for precise vascular navigation while aiming to reduce radiation exposure and physical strain. The Company commenced the Limited Market Release (LMR) of the LIBERTY system in late 2025 and plans for a Full Market Release (FMR) at the Society of Interventional Radiology (SIR) conference in April 2026, allowing the Company to showcase LIBERTY with the goal to deepen market adoption. About Microbot Medical Microbot Medical Inc. (NASDAQ: MBOT) is a commercial stage medical device company focused on transforming endovascular procedures through advanced robotic technology. Microbot’s LIBERTY® Endovascular Robotic System is the world’s first FDA cleared single-use, remotely operated robotic solution designed for precision, efficiency and safety. Backed by a strong intellectual property portfolio and a commitment to innovation, Microbot is driving the future of endovascular care. Learn more at www.microbotmedical.com and connect on LinkedIn and X. Safe Harbor Statements to future financial and/or operating results, future adoption of products, future growth in research, technology, clinical development, commercialization and potential opportunities for Microbot Medical Inc. and its subsidiaries, along with other statements about the future expectations, beliefs, goals, plans, or prospects expressed by management, constitute forward-looking statements within the meaning of the Private Securities Litigation Reform Act of 1995 and the Federal securities laws. Any statements that are not historical fact (including, but not limited to statements that contain words such as “contemplates,” “continues,” “could,” “forecasts,” “intends,” “may,” “might,” “possible,” “potential,” “predicts,” “projects,” “should,” “would,” “will,” “believes,” “plans,” “anticipates,” “expects,” “estimates” and similar expressions) should also be considered to be forward-looking statements, but the absence of these words does not mean that a statement is not forward-looking. Forward-looking statements involve risks and uncertainties, including, without limitation, market conditions, risks inherent in the commercialization of the LIBERTY® Endovascular Robotic System, and in the development of future versions of or applications for the system, uncertainty in the results of regulatory pathways and regulatory approvals, uncertainty resulting from political, social and geopolitical conditions, particularly any changes in personnel or processes or procedures at the FDA and announcements of tariffs on imports into the U.S., disruptions resulting from new and ongoing hostilities between Israel and the Palestinians, Iran and other neighboring countries, and maintenance of intellectual property rights. Additional information on risks facing Microbot Medical® can be found under the heading “Risk Factors” in Microbot Medical’s periodic reports filed with the Securities and Exchange Commission (SEC), which are available on the SEC’s web site at www.sec.gov. Microbot Medical® disclaims any intent or obligation to update these forward-looking statements, except as required by law. Contacts: IR@microbotmedical.comMedia@microbotmedical.com
Bracco Imaging and Canon Medical Systems Europe Announce Joint Symposium on CEUS at ECR 2026
Joint Session to Highlight the Evolving Role of Contrast-Enhanced Ultrasound in Modern Radiology Practice MILAN, March 4, 2026 /PRNewswire/ — Bracco Imaging S.P.A. today announced that it will co-host a scientific symposium with Canon Medical Systems Europe B. V. at the European Congress…
SHERPA research consortium initiates seven clinical studies to validate AI-based assistive technologies for minimally invasive brain and cancer treatments
Members of the SHERPA project team
Members of the SHERPA project team
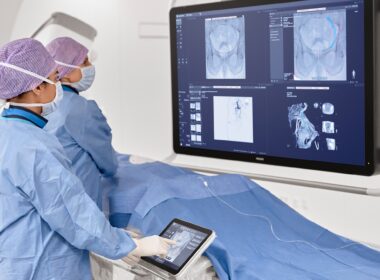
Philips Azurion interventional oncology procedure
Philips Azurion interventional oncology procedure
SHERPA project logo
SHERPA project logo
March 3, 2026 With a total budget of EUR 21.5 million, the four-year SHERPA research project is coordinated by Philips and co-funded by the EU Innovative Health Initiative (IHI) and industry partnersThe project focuses on developing automated workflows for minimally invasive neurovascular and tumor treatments – clinical procedures where staffing levels and specialized expertise are in short supplyThe clinical studies are being conducted at University Medical Center Utrecht, St Antonius Hospital, University Medical Center Hamburg-Eppendorf, Hôpital Bicêtre AP-HP, and Hospital de la Santa Creu i Sant PauOn March 4 (16:30 – 17:30 CET), SHERPA will be featured at the European Congress of Radiology 2026 in a dedicated session Amsterdam, the Netherlands – Royal Philips (NYSE: PHG, AEX: PHIA), a global leader in health technology, today announced that the Philips-coordinated SHERPA research consortium has initiated seven clinical studies to demonstrate the benefits of AI- and robotics-assisted workflows in minimally invasive treatments for brain aneurysms* and liver tumors. Staff shortages, coupled with the complex nature of the work, place significant pressure on interventional radiologists and interventional neuroradiologists who perform these procedures. The SHERPA consortium aims to validate AI-powered technologies in imaging, data visualization, procedure planning and guidance, clinical decision support, and patient pathway orchestration. These technologies are designed to automate repetitive, time-consuming tasks, support decision-making, and accelerate learning to ease the workload of interventional (neuro)radiologists. The four-year research project is co-funded by the industry partners through in-kind contributions and additional resources, as well as by the European Union Innovative Health Initiative (IHI), and comprises 16 partners from seven European countries. “Over the past year, the SHERPA project has brought together a talented team of researchers who have developed a technology framework designed to make workflow automation a reliable companion – a ‘sherpa’ – for interventional (neuro)radiologists as they navigate some of their most complex procedures,” said Bert van Meurs, Chief Business Leader, Image Guided Therapy, at Royal Philips. “By providing a template for the future adoption of AI-enabled assistive technologies and accelerating the associated learning curve, this framework will help address the increased demand for interventional (neuro)radiologists in Europe and beyond.” “Demand for image-guided, minimally invasive procedures has outpaced the growth of the specialized workforce,” said Irene van der Schaaf, Professor Neurovascular Interventional Radiology, UMC Utrecht. “At the same time, procedures are becoming more complex and technology is advancing rapidly, making it harder for teams to keep pace. In interventional radiology, technology and human touch go hand in hand. Physicians must manage highly technical tasks – sometimes away from the patient – while continuing to provide personal care and reassurance. The SHERPA project aims to develop AI-enabled tools that simplify routine tasks, enhance patient care, and support the training of the next generation of interventional (neuro)radiologists.” On March 4, 2026, from 16:30 to 17:30 CET, SHERPA will be featured at the European Congress of Radiology (ECR) 2026 in a dedicated session titled: “Assistive Technologies for Interventional Radiologists: Enhancing Decision-Making and Streamlining Workflow with Innovations and AI-Powered Solutions.” Click here to view the agenda and register to attend the session online. Innovation through public-private partnershipThe SHERPA public-private partnership consortium comprises five medical technology industry partners, five academic partners, and five research organization and medical association partners: Industry partners: Philips, Medtronic, Sim&Cure, Interventional Systems, Barco.Academic partners: University Medical Center Utrecht (The Netherlands), University Medical Center Hamburg-Eppendorf (Germany), Hôpital Bicêtre AP-HP (France), Hospital de la Santa Creu i Sant Pau (Spain), St Antonius Hospital (The Netherlands).Research organization and medical association partners: European Institute for Biomedical Imaging Research – EIBIR, Cardiovascular and Interventional Radiological Society of Europe – CIRSE, European Society of Minimally Invasive Neurological Therapy – ESMINT, Eindhoven University of Technology – TU/e (The Netherlands), Human-Factors-Consult – HFC (Germany). Addressing staff shortages and patient access by relieving the pressure on physiciansThe World Health Organization (WHO) predicts a shortage of 600 thousand physicians in the European Union (EU) by 2030 [1]. Some of the most acute physician shortages are in interventional radiology, even in countries such as the UK that have advanced healthcare systems [2]. Among the many different procedures conducted by interventional (neuro)radiologists, treatments of brain aneurysms and liver tumors are some of the most complex and time consuming. Both rely on CT and/or MR imaging for diagnosis, real-time imaging for procedure guidance, and a high level of precision in the placement of therapeutic devices such as blood-clot inducing platinum coils to seal off a brain aneurysm or the percutaneous insertion of ablation needles to treat a liver tumor. As a result, these procedures require a very high level of operator expertise and training, which can limit patient access. The SHERPA project aims to provide interventionists with AI-powered assistive technologies that automate repetitive tasks and support decision-making across the entire workflow. By doing so, they will accelerate learning curves and improve precision and safety in complex minimally invasive interventions such as brain aneurysm repair and liver tumor ablation. During the first year of the project, the consortium successfully developed AI algorithms to help identify brain aneurysms that need treatment and algorithms to optimize patient selection and therapy planning for liver tumor ablation. They also developed robotic technology to improve procedure precision and reduce difficulty, and AI software to confirm treatment success. These have now been integrated into orchestrated end-to-end workflows for both types of procedure. SHERPA project clinical studiesOver the next three years of the four-year SHERPA project, the consortium partners will conduct a series of clinical studies to refine these assistive technologies and assess the benefits in terms of the patient experience, workload optimization, interventionist satisfaction, and performance. The five brain aneurysm studies will focus on AI-driven aneurysm detection, risk prediction, and precise treatment planning: RADAR: AI-based aneurysm detection based on CT and MR imaging.Aneurysm@risk: AI-based algorithm to predict aneurysm growth and rupture risk.ASSIST: AI-supported device selection and positioning guidance.INTERACT: Automatic collimation and projection angle suggestions to optimize imaging for procedural guidance.SAFO: Evaluation of a digital remote follow-up solution for brain aneurysm patients, enabling standardized monitoring, enhanced coordination, and seamless care across the patient pathway. The liver and lung tumor studies will leverage advanced imaging and robotic-assisted biopsy technologies, respectively, to drive greater diagnostic precision and procedural efficiency. MISTRAL: Evaluation of new Cone Beam CT workflows to optimize imaging for percutaneous liver ablations.RHODES: Evaluation of robotic-assisted versus free-hand lung biopsies with a focus on operability and device efficiency. Together, these studies will generate insights to support the acceptance and adoption of AI-based smart assistive technologies by the wider interventional radiology community. The SHERPA website can be viewed at: https://sherpa-ihi.eu. Watch videos and interviews about SHERPA here. The CORDIS project page for SHERPA can be accessed here, and the IHI factsheet for SHERPA can be found here. Notes and references* A brain aneurysm is a weak, bulging spot in a blood vessel in the brain, which can leak or rupture.[1] Zapata T, Muscat NA, Falkenbach M, and Wismar M. WHO Report – From Great Attrition to Great Attraction: Countering the Great Resignation of Health and Care Workers. Eurohealth, Vol.29, No.1, 2023. https://iris.who.int/server/api/core/bitstreams/488b01ab-a066-4558-a345-476570fe2802/content[2] The Royal College of Radiologists: Clinical radiology – UK workforce census 2020 report. https://www.rcr.ac.uk/media/3gjdr23o/clinical_radiology_census_report_2020.pdf This project is supported by the Innovative Health Initiative Joint Undertaking (IHI JU) under grant agreement No 101194744. The JU receives support from the European Union’s Horizon Europe research and innovation program and life science industries represented by COCIR, EFPIA, Europa Bío, MedTech Europe and Vaccines Europe. SHERPA is Funded by the European Union. Views and opinions expressed are however those of the author(s) only and do not necessarily reflect those of the European Union or Innovative Health Initiative Joint Undertaking. Neither the European Union nor the granting authority can be held responsible for them. Media ContactSteve KlinkPhilips Medical and Scientific CommunicationsTel: +31 6 10888824E-mail: steve.klink@philips.com Joost MalthaPhilips Global External RelationsTel: +31 6 10558116E-mail: joost.maltha@philips.com About Royal PhilipsRoyal Philips (NYSE: PHG, AEX: PHIA) is a leading health technology company focused on improving people’s health and well-being through meaningful innovation. Philips’ patient- and people-centric innovation leverages advanced technology and deep clinical and consumer insights to deliver personal health solutions for consumers and professional health solutions for healthcare providers and their patients in the hospital and the home.Headquartered in the Netherlands, the company is a leader in diagnostic imaging, ultrasound, image-guided therapy, monitoring and enterprise informatics, as well as in personal health. Philips generated 2025 sales of EUR 18 billion and employs approximately 64,800 employees with sales and services in more than 100 countries. News about Philips can be found at www.philips.com/newscenter.
Attachments
Members of the SHERPA project team
Philips Azurion interventional oncology procedure
SHERPA project logo
Qure.ai Nets Six New Indications Cleared by the FDA, Taking the Chest X-ray Crown in the Radiology AI Race
NEW YORK–(BUSINESS WIRE)–Qure.ai nets six new indications cleared by the FDA, taking the chest X-ray crown in the radiology AI race
Bracco and the European Society of Radiology Empower the Future of Radiology at ECR 2026
Bracco to unveil its integrated MRI portfolio at the booth, alongside immersive experiences, scientific symposiums, and advances across MRI, CEUS, and AI in collaboration with the European Society of Radiology MILAN, Feb. 26, 2026 /PRNewswire/ — Bracco Imaging S.p.A., a global leader in…
Gentuity Announces Advanced Intravascular Imaging Solutions to Become Available Through GE HealthCare in the United States
Collaboration aims to expand access to High-Frequency OCT Imaging across U.S. cardiac catheterization labs SUDBURY, Mass., Feb. 25, 2026 /PRNewswire/ — Gentuity LLC, a Massachusetts-based medical technology company specializing in next-generation intravascular imaging devices, today…