WASHINGTON, June 24, 2019 /PRNewswire/ — The Centers for Medicaid and Medicare Services last Friday issued their final National Coverage Decision on how hospitals will be approved to do the TAVR (transaortic valve replacement) procedure, a less invasive valve replacement treatment that avoids open heart surgery and allows patients to resume daily life […]
Rhythm
Correvio Resubmits Brinavess (Vernakalant) New Drug Application to U.S. FDA for the Treatment of Patients with Recent Onset Atrial Fibrillation
VANCOUVER, June 24, 2019 /PRNewswire/ – Correvio Pharma Corp. (NASDAQ: CORV) (TSX: CORV), a specialty pharmaceutical company focused on commercializing hospital drugs, today announced it has resubmitted a New Drug Application (NDA) to the U.S. Food and Drug Administration (FDA) seeking approval for Brinavess™ (vernakalant hydrochloride, IV), its antiarrhythmic drug for the rapid conversion […]
Philips receives FDA premarket approval for its HeartStart OnSite and HeartStart Home defibrillators
Amsterdam, the Netherlands – Royal Philips (NYSE: PHG, AEX: PHIA), a global leader in health technology, today announced that the Center for Devices and Radiological Health (CDRH) of the Food and Drug Administration (FDA) has approved the company’s premarket approval (PMA) application for its HeartStart OnSite defibrillator [1] and HeartStart Home defibrillator [2], […]
PAI Health Joins American Heart Association Innovators Network to Scale Digital Solutions that Improve Cardiovascular Health
VANCOUVER, British Columbia, June 18, 2019 (GLOBE NEWSWIRE) — PAI Health, a heart health software company, will join the American Heart Association’s Center for Health Technology & Innovation (CHTI) Innovators Network to contribute their science-based technology that helps reduce the risk of cardiovascular disease, the leading cause of death for all […]
Stereotaxis Announces the First Graduating Class of the Robotic Electrophysiology Fellows Program
ST. LOUIS, June 18, 2019 (GLOBE NEWSWIRE) — Stereotaxis, Inc. (OTCQX: STXS), the global leader in innovative robotic technologies for the treatment of cardiac arrhythmias, today announced the first graduating class from its Robotic Electrophysiology Fellows Program. The Robotic Electrophysiology Fellows Program is designed to enhance the traditional electrophysiology fellowship […]
CardioFocus Launches 100th HeartLight® Center Worldwide
MARLBOROUGH, Mass., June 18, 2019 /PRNewswire/ — CardioFocus, Inc., a medical device company dedicated to advancing ablation treatment for atrial fibrillation (AFib), announced today that the HeartLight® Endoscopic Ablation System is now offered at more than 100 hospitals worldwide. The HeartLight System is a revolutionary catheter ablation technology for controlled and consistent pulmonary vein isolation (PVI), the […]
Aurora Resurgence Announces Sale of Cardiac Science Corporation to ZOLL Medical Corporation
LOS ANGELES, June 11, 2019 /PRNewswire/ — Aurora Resurgence, a Los Angeles-based private equity firm, today announced that its affiliate has entered into a definitive agreement to sell Cardiac Science Corporation (“Cardiac Science” or “CSC”), a leading provider of automated external defibrillators (“AEDs”), related services and accessories, to ZOLL® Medical Corporation, an Asahi Kasei […]
Zoll Introduces New Technology to Improve the Management of Acute Heart Failure Patients
CHELMSFORD, Mass.–(BUSINESS WIRE)–ZOLL® Medical Corporation, an Asahi Kasei Group company that manufactures medical devices and related software solutions, announced the launch of and first patient prescription for the ZOLL µCor™ (pronounced “Micro Core”) Heart Failure and Arrhythmia Management System (HFAMS), designed to help clinicians improve outcomes and reduce hospitalizations for […]
BioSig Allowed a U.S. Patent for PURE EP™ Simulator
Santa Monica, CA, June 06, 2019 (GLOBE NEWSWIRE) — BioSig Technologies, Inc. (NASDAQ: BSGM), a medical device company developing a proprietary biomedical signal processing platform designed to address an unmet technology need for the electrophysiology (EP) marketplace, today announced that the U.S. Patent & Trademark Office allowed a U.S. patent application covering its […]
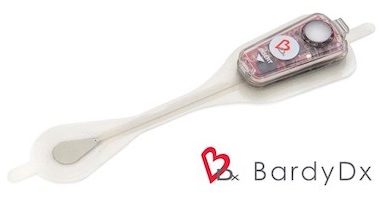
Bardy Diagnostics™ Selected as Winner of the MedTech Breakthrough Award For Best New Diagnostic Technology
SEATTLE, June 6, 2019 /PRNewswire/ — Bardy Diagnostics, Inc., (“BardyDx”), a leading provider of ambulatory cardiac monitoring technologies and custom data solutions, announced that MedTech Breakthrough, an independent organization that recognizes the top companies and solutions in the global health and medical technology market, has recognized the Carnation Ambulatory Monitor (CAM™) with the […]