AUSTIN, Texas, June 4, 2019 /PRNewswire/ — The Texas Cardiac Arrhythmia Institute (TCAI) at St. David’s Medical Center is opening a new, state-of-the-art Electrophysiology Center dedicated to treating patients with cardiac arrhythmias. The new Electrophysiology Center is set to open at the end of the month. “This expansion at the Texas Cardiac Arrhythmia Institute […]
Rhythm
touchCARDIO Joins Forces with Arrhythmia Alliance
LONDON, June 5, 2019 /PRNewswire/ — This World Heart Rhythm Week (3-9 June 2019) Arrhythmia Alliance has announced a collaboration with touchCARDIO that will see the two join forces to further improve the diagnosis, treatment and quality of life for all those affected by arrhythmias. Arrhythmia Alliance World Heart Rhythm Week is an […]
BioTrace Medical Announces Key Events Featuring Tempo Temporary Pacing Lead at TVT 2019
MENLO PARK, Calif.–(BUSINESS WIRE)–BioTrace Medical, Inc., the leader in innovative temporary pacing technology, today announced the company’s key activities at the Annual Transcatheter Valve Therapy Structural Heart Summit (TVT 2019), which will take place June 12-15 at the Sheraton Grand Chicago. The company’s Tempo® Temporary Pacing Lead will be featured in […]
Medtronic Gains U.S. FDA Clearance for New Catheter System for HIS Bundle Pacing
DUBLIN – May 29, 2019 – Medtronic plc (NYSE:MDT) today announced U.S. Food and Drug Administration (FDA) clearance and commercial launch for the SelectSite(TM) C304-HIS deflectable catheter system for use in procedures involving His-bundle pacing (HBP). The SelectSite C304-HIS deflectable catheter system features a deflectable, out-of-plane curve to reach the bundle […]
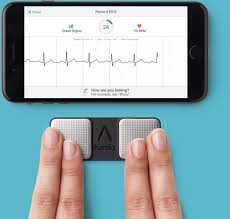
AliveCor and Best Buy Partner to Bring KardiaMobile to Select Stores Nationwide
MOUNTAIN VIEW, Calif., May 29, 2019 /PRNewswire/ — AliveCor, the leader in FDA-cleared consumer electrocardiogram (ECG) technology, and Best Buy Co., Inc. (NYSE: BBY) today announced a strategic partnership, bringing AliveCor’s products to select Best Buy stores across the country. This is the first time AliveCor’s products will be sold in consumer electronics stores […]
Orchestra BioMed™ Presents Clinical Data at EuroPCR 2019 Demonstrating Safety and Efficacy of BackBeat Cardiac Neuromodulation Therapy (CNT)
Novel implantable bioelectronic treatment delivers immediate, substantial, and sustained reduction in blood pressure Data showed 14.2 mmHg reduction in 24-hour ambulatory systolic blood pressure at three months and 23.4 mmHg reduction in office cuff systolic blood pressure out to two years Therapy applicable to a wide range of hypertensive patients […]
BioSig Allowed First Key US Patent Claims for its PURE EP™ System
Santa Monica, CA, May 23, 2019 (GLOBE NEWSWIRE) — BioSig Technologies, Inc. (NASDAQ: BSGM), a medical device company developing a proprietary biomedical signal processing platform designed to address an unmet technology need for the electrophysiology (EP) marketplace, today announced that the US Patent Office allowed 33 patent claims covering its PURE EP™ System. […]
Boston Scientific Initiates Trial Comparing Left Atrial Appendage Closure to Direct Oral Anticoagulants for Stroke Risk Reduction Post-AFib Ablation
MARLBOROUGH, Mass., May 22, 2019 /PRNewswire/ — Boston Scientific Corporation (NYSE: BSX) has initiated the OPTION trial to compare safety and effectiveness of the next-generation WATCHMAN FLX™ left atrial appendage closure (LAAC) platform to first-line oral anticoagulants (OAC) – including direct oral anticoagulants (DOAC) and warfarin – for stroke risk reduction in patients with […]
Results Announced from Two Renal Denervation Studies at EuroPCR Show Positive Outcomes in High-Risk Hypertensive Patients
DUBLIN and PARIS – May 22, 2019 – Investigators today unveiled late-breaking clinical data from a first-of-its-kind physician sponsored clinical trial. The data indicate that renal denervation (RDN) with the Medtronic Symplicity(TM) renal denervation system was associated with reduced occurrence of subclinical atrial fibrillation (AF) in a small subset of high-risk […]
Hemovent Receives CE Marking for MOBYBOX™ ECLS System
AACHEN, Germany–(BUSINESS WIRE)–Hemovent GmbH announced today that it has received CE Marking for its MOBYBOX™ ECLS System, allowing the company to market its innovative product throughout the European Union. MOBYBOX is a miniaturized heart/lung machine that uses Hemovent’s proprietary Bionique Flow Technologies as its platform. As the world’s first self-contained and fully integrated […]