by Ken Dropiewski, Prime-Core Executive Search (ken@prime-core.com) It has the makings of a plot for a blockbuster movie: a high ranking government official or world leader assassinated by a nefarious hacker who remotely accesses an implanted cardiac device. Plot lines and prominent world leaders notwithstanding, cyber-security risks for implantable cardiac […]
Rhythm
Cardiome Provides U.S. Regulatory Update For BRINAVESS
VANCOUVER, Aug. 21, 2017 /PRNewswire/ – Cardiome Pharma Corp. (NASDAQ: CRME/ TSX:COM) today announced that it has received a response from the U.S. Food and Drug Administration (FDA) regarding the regulatory path for BRINAVESS® (vernakalant hydrochloride, IV), the Company’s antiarrhythmic drug for the rapid conversion of recent onset atrial fibrillation (AF). In its […]
BIOTRONIK US Launches Smallest MR Conditional Quadripolar Cardiac Resynchronization Therapy Pacemaker
LAKE OSWEGO, Ore., Aug. 21, 2017 /PRNewswire/ — BIOTRONIK today announced FDA approval and commercial availability of Edora HF-T QP, an MR conditional quadripolar (QP) cardiac resynchronization therapy pacemaker (CRT-P) with MRI AutoDetect technology. With a volume of 15 cc, Edora HF-T QP is the smallest MR conditional CRT-P available in the US with […]
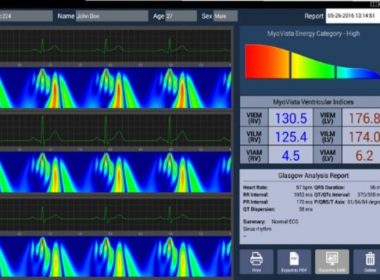
HeartSciences Announces CE Mark and European Launch of MyoVista® High Sensitivity ECG Device
WESTLAKE, Texas, Aug. 17, 2017 /PRNewswire/ — HeartSciences today announced the European launch of MyoVista® high sensitivity electrocardiograph (hsECG™) Testing Device, developed in response to the global unmet need for effective, low-cost, front-line screening of cardiac disease in both symptomatic and asymptomatic patients. MyoVista measures the heart’s energy during each […]
AUM CARDIOVASCULAR RECEIVES FDA CLEARANCE FOR REVOLUTIONARY DIAGNOSTIC HEART DEVICE
NORTHFIELD, MINNESOTA— AUM Cardiovascular announced that it has received clearance from the Food and Drug Administration for CADence,™ a non-invasive acoustic and ECG device designed to help physicians detect physiological and pathological heart murmurs. The reusable, non-invasive, radiation-free handheld device, which is now available in the United States, records sounds […]
Boca Raton Regional Hospital Using New Technology To Map Irregular Heart Rhythms Non-Invasively
NEWS PROVIDED BY Boca Raton Regional Hospital Aug 01, 2017, 12:24 ET BOCA RATON, Fla., Aug. 1, 2017 /PRNewswire-USNewswire/ — Boca Raton Regional Hospital is the first in Florida and one of only five centers nationally to non-invasively map irregular heartbeats in patients with chronic atrial fibrillation and other arrhythmias who have not responded adequately […]
CardioFocus® Receives Approval For HeartLight® Endoscopic Ablation System From The Japanese Ministry Of Health, Labour And Welfare
MARLBOROUGH, Mass., July 31, 2017 /PRNewswire/ — CardioFocus, Inc. today announced that the Japanese Ministry of Health, Labour and Welfare has approved the HeartLight Endoscopic Ablation System for the treatment of paroxysmal atrial fibrillation (AF) in Japan. The HeartLight System is a visually guided laser balloon technology for controlled and consistent pulmonary vein isolation (PVI) […]
Allied Minds’ Portfolio Company Signature Medical Raises $2.5 Million in Series A Financing
BOSTON–(BUSINESS WIRE)–Signature Medical, Inc. (Signature Medical), an Allied Minds (LSE: ALM) portfolio company, today announced that it has completed a $2.5 million Series A funding round, including participation from Riot Ventures, an early stage investment group focused on emerging technologies; Bose Corporation, a global leader in audio innovation; and Allied […]
Single Scenario Causes Death for Boston Scientific’s S-ICD
Boston Scientific warns on fluke S-ICD death JULY 28, 2017 BY BRAD PERRIELLO , MassDevice Boston Scientific (NYSE:BSX) last month alerted physicians after learning of a fluke incident involving its S-ICD pacemaker, in which a patient died when the device’s memory was corrupted by radiation. In a June letter to physicians, Marlborough, Mass.-based Boston […]
Researchers Prove Human Heart’s ‘Battery’ Has Multiple Backups
Newswise — COLUMBUS, Ohio – There’s good news when it comes to our heart’s sinoatrial node (SAN), the body’s natural pacemaker. Scientists at The Ohio State University Wexner Medical Center have shown the human SAN is hardwired with a backup system — three diverse regions of pacemakers acting as batteries and up […]