SAN FRANCISCO, July 10, 2017 (GLOBE NEWSWIRE) — iRhythm Technologies, Inc. (NASDAQ:IRTC), a leading digital health care solutions company focused on the advancement of cardiac care announced a collaboration with the Stanford Machine Learning Group that has resulted in the development of a deep learning algorithm capable of expert level […]
Rhythm
BioSig Technologies Enters Into Strategic Development Work With Health Research International
Minneapolis, MN, July 11, 2017 (GLOBE NEWSWIRE) — BioSig Technologies, Inc. (OTCQB: BSGM), a medical device company developing a proprietary platform designed to address an unmet technology need within the $4+ billion electrophysiology (EP) marketplace, today announced that the Company has engaged Health Research International (HRI) to compile essential market […]
Could Our Vehicles be First Responders in the Near Future?
By Ken Dropkiewski (ken@prime-core.com) While no hard statistics exist as to the exact number of motor vehicle accidents and concurrent heart attacks, the article 5 Facts About the Trend of Heart Attacks While Driving, makes a compelling case for why more people are suffering acute MIs while driving. – An aging population […]
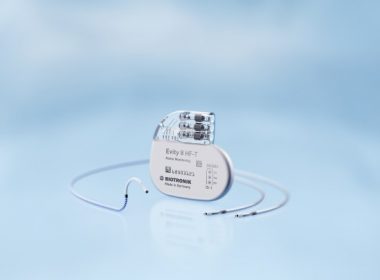
BIOTRONIK’s New Quadripolar CRT-P with More Pacing Options Now Available in Japan
Evity 8 HF-T QP Offers a Host of Functions Including LV VectorOpt, RF Telemetry, MRI AutoDetect and BIOTRONIK Home Monitoring TOKYO, Japan and BERLIN, Germany, July 10, 2017 – BIOTRONIK has announced the launch of its Evity cardiac resynchronization therapy pacemaker (CRT-P) in the Japanese market. It is the company’s highest performing […]
CathVision Raises Venture Funding to Market New EP Recording System
COPENHAGEN, Denmark, July 7, 2017 /PRNewswire/ — Medical device company CathVision has signed a multi-million venture investment from Scandinavian-based investors VF Venture and Borean Innovation. The investment is directed at developing and market the company’s superior electrophysiology (EP) recording system for treating cardiac arrhythmia. Approximately 2% of the Western population suffers from cardiac […]
“Cardiologs ECG Analysis Platform” Receives FDA Clearance
PARIS–(BUSINESS WIRE)–Cardiologs Technologies SAS announced today that it has received FDA clearance of its Cardiologs ECG Analysis Platform, a cloud-based cardiac monitoring-analysis web service powered by artificial intelligence (AI). Cardiologs aids physicians in screening for atrial fibrillation (AFib) and other arrhythmias using long-term ambulatory ECG monitoring recordings. The Cardiologs system is also CE-marked […]
CardioFocus Announces Initiation Of Three Major Studies Featuring The HeartLight System For The Treatment Of Atrial Fibrillation
MARLBOROUGH, Mass., June 29, 2017/PRNewswire/ — CardioFocus, Inc., a medical device innovator and manufacturer dedicated to advancing ablation treatment for cardiac disorders such as atrial fibrillation (AF), today announced that its HeartLight Endoscopic Ablation System is being featured in three new major clinical studies currently underway. Collectively, up to 1,000 AF patients will be […]
United States Recovers $13.4 Million in False Claims Act Case for Medically Unnecessary Cardiac Monitoring Services
NEWS PROVIDED BY Whistleblower Law Collaborative 26 Jun, 2017, 15:37 ET BOSTON, June 26, 2017 /PRNewswire/ — Attorneys Suzanne E. Durrell and Robert M. Thomas, Jr. of the Whistleblower Law Collaborative are pleased to announce that the United States has settled a federal False Claims Act case brought by their whistleblower client, Eben Steele, against Medi-Lynx Cardiac Monitoring, LLC, AMI […]
BioSig Technologies Release: Novel Findings Obtained With The PURE EP System To Be Presented At American Heart Association’s BCVS Scientific Sessions 2017
Minneapolis, MN, June 26, 2017 (GLOBE NEWSWIRE) — BioSig Technologies, Inc. (OTCQB: BSGM), a medical device company developing a proprietary platform designed to address an unmet technology need for the $4+ billion electrophysiology (EP) marketplace, today announced that the American Heart Association’s 13th Annual Basic Cardiovascular Sciences (BCVS) 2017 Scientific […]
Millar, Inc. Sells Inca® Pressure-Volume (PV) Loop System to the University of Colorado to Support LVAD Research
HOUSTON, June 22, 2017 /PRNewswire/ — Millar, Inc. is pleased to announce that the University of Colorado Anschutz Medical Campus has purchased the CD Leycom® Inca® Pressure-Volume (PV) Loop System – sold in North America exclusively by Millar, Inc. – to better understand how left ventricular assist devices (LVADs) impact ventricular function […]