Heart failure is a global pandemic with an estimated 64.3 million cases worldwide in 2017, costing over US$100B per year Drug developers currently lack effective humanized models for preclinical testing of targeted drug candidates in heart failure with preserved ejection fraction The collaboration aims to provide a unique solution for […]
Other News
Mesoblast Financial Results for the Quarter Ended September 30, 2019
Continued Increase in Revenues and Strong Balance Sheet MELBOURNE, Australia and NEW YORK, Nov. 25, 2019 (GLOBE NEWSWIRE) — Mesoblast Limited (ASX:MSB; Nasdaq:MESO) today reported operational highlights and financial results for the first quarter ended September 30, 2019. Mesoblast Chief Executive Dr Silviu Itescu stated: “The Company’s financial results for […]
Resverlogix Announces Warrant Exercise Incentive Program
CALGARY, Alberta, Nov. 25, 2019 (GLOBE NEWSWIRE) — Resverlogix Corp. (“Resverlogix” or the “Company”) (TSX: RVX) announces a warrant exercise incentive program (“Incentive Program”) designed to encourage the early exercise of 15,593,428 warrants to purchase common shares of the Company. The Incentive Program does not apply to warrants held by […]
Acasti Pharma Reports Last Patient Visit in TRILOGY 1 Phase 3 Trial of CaPre for the Treatment of Severe Hypertriglyceridemia
On track to report topline results for TRILOGY 1 in December 2019 and TRILOGY 2 in January 2020 LAVAL, Québec, Nov. 26, 2019 (GLOBE NEWSWIRE) — Acasti Pharma Inc. (“Acasti” or the “Company”) (NASDAQ: ACST – TSX-V: ACST), a biopharmaceutical innovator focused on the research, development and commercialization of its […]
Itamar™ Medical Reports Record Third Quarter 2019 Revenues
– Third Quarter Revenues Increase 33% to $8.1 Million – – WatchPAT™ Revenues Increase 32% in the Third Quarter to $7.6 Million – – Company to Host Conference Call Today at 8:00 am ET, 3:00 pm IT – CAESAREA, Israel, Nov. 26, 2019 (GLOBE NEWSWIRE) — Itamar Medical Ltd. (Nasdaq: […]
OTITOPIC™ New Clinical Trial Results Continue to Demonstrate Inhibition of Platelet Aggregation in Two Minutes With Dry Powder Inhalation of Aspirin Formulation for High Risk Patients to Treat Suspected Acute Myocardial Infarction (MI)
LOS ANGELES–(BUSINESS WIRE)–OTITOPIC – A clinical-stage dry powder inhalation of aspirin pharmaceutical company pioneering the development of antithrombotic treatments for use at the time of suspected acute myocardial infarction (MI), today announced the successful conclusion based on PD results of a clinical trial for its lead drug: Dry Powder Inhalation […]
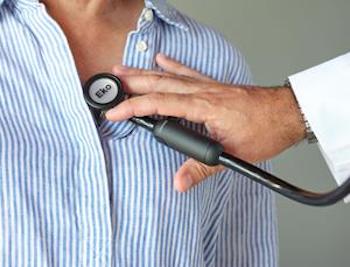
Eko Makes Stethoscopes Even Smarter with the Redesigned CORE
The redesigned Eko CORE boasts active noise cancellation, sleeker design, and powerful software to allow medical professionals to more effectively screen for heart disease San Francisco, CA, Nov. 25, 2019 (GLOBE NEWSWIRE) — Eko, a digital health company with an AI-powered cardiac screening platform, today announced the second generation of its CORE […]
Novartis to acquire The Medicines Company for USD 9.7 bn, adding inclisiran, a potentially transformational investigational cholesterol-lowering therapy to address leading global cause of death
Over 50 million secondary prevention patients worldwide with atherosclerotic cardiovascular disease (ASCVD) or familial hypercholesterolemia (FH) on current standard of care do not achieve LDL cholesterol (LDL-C) goal and remain at increased risk of cardiovascular events1 First-in-class siRNA biological mechanism enables unique twice-yearly, subcutaneous dosing regimen and seamless integration into […]
JanOne Acquires Worldwide, Exclusive License for Promising Treatment of Peripheral Arterial Disease (PAD)
Phase 2b testing planned for PAD treatment and PAD-associated pain to address 8.5 million patient US market LAS VEGAS, Nov. 25, 2019 /PRNewswire/ — JanOne Inc. (NASDAQ: JAN), a company focused on reducing opioid addiction by finding treatments for conditions accompanied by pain and bringing to market drugs and therapies with nonaddictive pain-relieving properties, […]
Favorable Outcomes with TCAR vs. Carotid Endarterectomy in Patients with Carotid Artery Disease
Updated Results of TCAR Surveillance Project Presented at 2019 VEITHsymposium NEW YORK, New York – November 22, 2019 – Silk Road Medical, Inc. (Nasdaq: SILK), a company focused on reducing the risk of stroke and its devastating impact, today announced the presentation of real-world data for the treatment of […]