LOS ANGELES–(BUSINESS WIRE)–OTITOPIC – A clinical-stage dry powder inhalation of aspirin pharmaceutical company pioneering the development of antithrombotic treatments for use at the time of suspected acute myocardial infarction (MI), today announced the successful conclusion based on PD results of a clinical trial for its lead drug: Dry Powder Inhalation […]
Other News
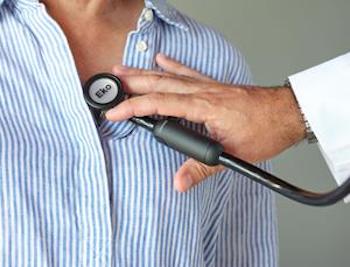
Eko Makes Stethoscopes Even Smarter with the Redesigned CORE
The redesigned Eko CORE boasts active noise cancellation, sleeker design, and powerful software to allow medical professionals to more effectively screen for heart disease San Francisco, CA, Nov. 25, 2019 (GLOBE NEWSWIRE) — Eko, a digital health company with an AI-powered cardiac screening platform, today announced the second generation of its CORE […]
Novartis to acquire The Medicines Company for USD 9.7 bn, adding inclisiran, a potentially transformational investigational cholesterol-lowering therapy to address leading global cause of death
Over 50 million secondary prevention patients worldwide with atherosclerotic cardiovascular disease (ASCVD) or familial hypercholesterolemia (FH) on current standard of care do not achieve LDL cholesterol (LDL-C) goal and remain at increased risk of cardiovascular events1 First-in-class siRNA biological mechanism enables unique twice-yearly, subcutaneous dosing regimen and seamless integration into […]
JanOne Acquires Worldwide, Exclusive License for Promising Treatment of Peripheral Arterial Disease (PAD)
Phase 2b testing planned for PAD treatment and PAD-associated pain to address 8.5 million patient US market LAS VEGAS, Nov. 25, 2019 /PRNewswire/ — JanOne Inc. (NASDAQ: JAN), a company focused on reducing opioid addiction by finding treatments for conditions accompanied by pain and bringing to market drugs and therapies with nonaddictive pain-relieving properties, […]
Favorable Outcomes with TCAR vs. Carotid Endarterectomy in Patients with Carotid Artery Disease
Updated Results of TCAR Surveillance Project Presented at 2019 VEITHsymposium NEW YORK, New York – November 22, 2019 – Silk Road Medical, Inc. (Nasdaq: SILK), a company focused on reducing the risk of stroke and its devastating impact, today announced the presentation of real-world data for the treatment of […]
Heartseed Raises $26M in Series B Funding to Accelerate Development of iPSC-derived Regenerative Medicine for Heart Failure
TOKYO–(BUSINESS WIRE)–Heartseed (Heartseed Inc.), a Tokyo-based biotechnology company developing iPSC-derived cardiomyocytes for the treatment of heart failure (HF), today announced it has raised 2.8 Billion-yen (approx. $26 Million) at Series B round, bringing its total financial backing to 3.8 Billion yen (approx. $35 Million) since it was founded in 2015. […]
Stereotaxis CEO to Present at the 12th Annual BIOMEDevice San Jose Conference
Conference Focuses on Role of Digital Technology in Healthcare; David Fischel Named Panelist in Robotics Session ST. LOUIS, Nov. 22, 2019 (GLOBE NEWSWIRE) — Stereotaxis (NYSE: STXS), the global leader in innovative robotic technologies for the treatment of cardiac arrhythmias, announced today its Chairman and Chief Executive Officer, David Fischel, will present […]
PAVmed Reports Third Quarter 2019 Financial Results and Provides Business Update
Conference call to be held on November 21, 2019 at 4:30 p.m. Eastern time NEW YORK, Nov. 21, 2019 (GLOBE NEWSWIRE) — PAVmed Inc. (Nasdaq: PAVM, PAVMZ) (the “Company” or “PAVmed”), a highly differentiated, multiproduct medical device company, today reported financial results for the three and nine months ended September […]
Neovasc Reducer™ Featured During Therapy Development Program in Coburg and Session at Maria Cecilia Hospital in Cotignola
VANCOUVER, CA, COBURG, DE, and COTIGNOLA, IT, Nov. 22, 2019 (GLOBE NEWSWIRE) — via NEWMEDIAWIRE — Neovasc, Inc. (“Neovasc” or the “Company”) (NASDAQ, TSX: NVCN), a leader in the development of minimally invasive transcatheter mitral valve replacement technologies and in the development of minimally invasive devices for the treatment of refractory angina, announced […]
Data Show Zilver® PTX® Leads to Fewer Complications and Shorter Hospital Stays Than Traditional Bypass Surgery
BLOOMINGTON, Ind.–(BUSINESS WIRE)–At this year’s VEITHsymposium®, Dr. Marc Bosiers presented data that show that patient treatment with the Zilver® PTX® stent has several benefits when compared to traditional bypass surgery. The data, which were gathered from a randomized controlled trial, show that treatment with Zilver PTX results in fewer complications and shorter […]