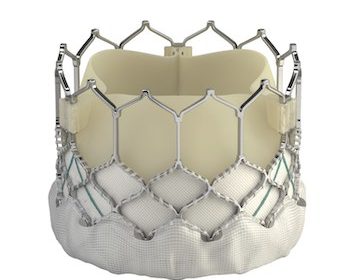
Superior TAVI Valve Indicated for All Patients Diagnosed with Aortic Stenosis IRVINE, Calif., Nov. 6, 2019 /PRNewswire/ — Edwards Lifesciences Corporation (NYSE: EW), the global leader in patient-focused innovations for structural heart disease and critical care monitoring, today announced that it has received CE Mark to expand use of the Edwards SAPIEN 3 […]
Other News
Impulse Dynamics Secures Transitional Pass-through (TPT) Incremental Payment for the Optimizer Smart System
2020 Payment Rules from the Centers for Medicare and Medicaid Services Bring CCM Therapy to Heart Failure Patients in the Outpatient Setting MOUNT LAUREL, N.J.–(BUSINESS WIRE)–Impulse Dynamics, developer of the implantable Optimizer Smart System for delivering Cardiac Contractility Modulation (also known as CCM™ therapy), announced that it has secured transitional […]
Neuroradiologist in Toronto Performs World’s First Robotic-Assisted Aneurysm Coiling Using Corindus Technology
Novel procedure marks a pivotal step in implementation of robotics to address neurovascular disease TORONTO & WALTHAM, Mass.–(BUSINESS WIRE)–Corindus, a Siemens Healthineers Company and leading developer of precision vascular robotics, announced today its CorPath® GRX System was used to complete the first-in-human robotic-assisted neurovascular intervention. The procedure was performed by the […]
CHF Solutions, Inc. Announces Third Quarter Financial Results and Provides Company Update
EDEN PRAIRIE, Minn., Nov. 05, 2019 (GLOBE NEWSWIRE) — CHF Solutions, Inc. (Nasdaq: CHFS), a medical device company focused on developing, manufacturing and commercializing a clinically proven solution for diuretic-resistant patients suffering from fluid overload, announced today its results for the third quarter ended September 30, 2019, which included the […]
MyoKardia Reports Third Quarter 2019 Financial Results
SOUTH SAN FRANCISCO, Calif., Nov. 04, 2019 (GLOBE NEWSWIRE) — MyoKardia, Inc. (Nasdaq: MYOK), today reported financial results for the quarter ended September 30, 2019. “As we look across the milestones recently achieved and those just ahead, we see steady progress on our near- and long-term corporate objectives. We are […]
Amarin Reports Third Quarter 2019 Financial Results and Provides Update on Operations
Record Revenue Levels and Preparations On-Track for Further Expansion Management to Host Conference Call at 7:30 a.m. ET Today DUBLIN, Ireland and BRIDGEWATER, N.J., Nov. 05, 2019 (GLOBE NEWSWIRE) — Amarin Corporation plc (NASDAQ:AMRN), a pharmaceutical company focused on improving cardiovascular health, today announced financial results for the three and nine […]
Okami Medical Announces Major Milestones: FDA 510(k) Clearance And Key Patent For The LOBO Vascular Occluder
Microcatheter-delivered device designed to provide rapid and focal occlusion of a wide range of peripheral arterial targets ALISO VIEJO, Calif., Nov. 4, 2019 /PRNewswire/ — Okami Medical Inc., a medical device company, today announced 510(k) clearance by the U.S. Food and Drug Administration (FDA) and initial launch of the LOBOTM Vascular Occlusion System. […]
CORRECTING and REPLACING PHOTO PQ Bypass Initiates Pivotal Study of Next-Generation Advancement in Stent Graft Technology for Treatment of Patients with Peripheral Artery Disease
MILPITAS, Calif.–(BUSINESS WIRE)–Please replace the photo with the accompanying corrected photo. The release reads: PQ Bypass, Inc., a clinical-stage medical device company, today announced it has enrolled the first patient in its TORUS 2 multi-center clinical trial of its self-expanding TORUS® stent graft system, a novel technology designed for the […]
GE Healthcare Announces U.S. FDA Approval of Macrocyclic MRI Contrast Agent Clariscan™ (gadoterate meglumine) Injection for Intravenous Use
Clariscan, approved in more than 55 countries globally with over four million patient doses shipped, is now FDA-approved in the U.S. Expands the GE Healthcare portfolio of contrast media products CHALFONT ST GILES, England–(BUSINESS WIRE)–The U.S. Food and Drug Administration (FDA) has approved Clariscan™, a macrocyclic, ionic, gadolinium-based, MRI contrast agent, […]
Shockwave Announces Commercial Availability of Below-the-Knee IVL Catheter
Shockwave S4 Catheter Expands the Intravascular Lithotripsy System Portfolio to Address the Challenges of Critical Limb Ischemia SANTA CLARA, Calif., Nov. 04, 2019 (GLOBE NEWSWIRE) — Shockwave Medical, Inc. (NASDAQ: SWAV), a pioneer in the development of Intravascular Lithotripsy (IVL) to treat complex calcified cardiovascular disease, today announced commercial availability of […]