VANCOUVER, Nov. 4, 2019 /PRNewswire/ – Correvio Pharma Corp. (NASDAQ: CORV) (TSX: CORV), a specialty pharmaceutical company focused on commercializing hospital drugs, today announced that the U.S. Food and Drug Administration (FDA) will hold a Cardiovascular and Renal Drugs Advisory Committee (CRDAC) meeting in connection with its review of the pending New Drug […]
Other News
Cook Medical India Opens New Facility in Chennai
CHENNAI, India–(BUSINESS WIRE)–Cook Medical India celebrated its new office opening. The company has shifted to a modern, 20,000-square foot office at Kochar Jade in Guindy, Chennai. It is located at the city’s major business district with easy access to land and air freight stations. India is an important part of […]
Ra Medical Systems Announces Substantial Completion of Audit Committee Investigation
CARLSBAD, Calif.–(BUSINESS WIRE)–Ra Medical Systems, Inc. (NYSE: RMED), a medical device company focusing on commercializing excimer laser systems to treat vascular and dermatological diseases, today announced that the Audit Committee of the Board of Directors (the “Audit Committee”) has substantially completed its internal investigation, which was originally announced in August. […]
SignPath Pharma, Inc. Announces an Out-license Agreement of Its CorreQTTM Technology for Mitigation of QT Prolongation with Rain Therapeutics Inc.
SALT LAKE CITY–(BUSINESS WIRE)–SignPath Pharma (Salt Lake City UT) today announced the first commercial out-license of its proprietary CorreQT technology platform. SignPath entered into an agreement with Rain Therapeutics (Newark CA), which licenses SignPath’s CorreQT technology for use in conjunction with Rain’s proprietary lead compound, Tarlox® (tarloxotinib,) a potent pan-ErbB […]
Proteon Therapeutics Announces Third Quarter 2019 Financial Results
WALTHAM, Mass., Oct. 31, 2019 (GLOBE NEWSWIRE) — Proteon Therapeutics, Inc. (“Proteon” or the “Company”) (Nasdaq: PRTO), a company that has historically focused on the development of novel, first-in-class pharmaceuticals to address the medical needs of patients with kidney and vascular disease, today announced its financial results for the quarter ended September […]
Stereotaxis and ADAS 3D Combine Robotic Precision and Advanced Preoperative Substrate Mapping to Treat Cardiac Arrhythmias
ST. LOUIS and BARCELONA, Spain, Nov. 01, 2019 (GLOBE NEWSWIRE) — Stereotaxis (NYSE American: STXS) and ADAS 3D Medical SL today announced the first patients have been successfully treated with the integration of ADAS 3D’s advanced preoperative substrate mapping and Stereotaxis’ Robotic Magnetic Navigation technologies. ADAS 3D helps identify possible sources of dangerous irregular heartbeats and […]
Neovasc to Submit Full PMA Application for Neovasc Reducer™ in 2019
VANCOUVER, Nov. 01, 2019 (GLOBE NEWSWIRE) — via NEWMEDIAWIRE — Neovasc, Inc. (“Neovasc” or the “Company”) (NASDAQ, TSX: NVCN), a leader in the development of minimally invasive transcatheter mitral valve replacement technologies and in the development of minimally invasive devices for the treatment of refractory angina, today announced that it intends to submit […]
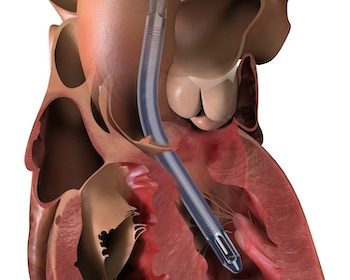
First U.S. Patients Treated with Impella 5.5 with SmartAssist, a Minimally Invasive, Forward Flow Heart Pump
DANVERS, Mass.–(BUSINESS WIRE)–Three cardiac surgeons at Cleveland Clinic, Hackensack Meridian Health and Cedars-Sinai Medical Center are the first in the United States to implant Abiomed’s (NASDAQ: ABMD) newest heart pump, the Impella 5.5 with SmartAssist. Ed Soltesz, MD, Mark Anderson, MD, and Danny Ramzy, MD, have each successfully implanted multiple pumps during cardiac procedures at their hospitals. Mark […]
myTAIHEART Test Provides Evidence for Injury from Biopsy of Heart Transplant Recipients
MILWAUKEE, Wis.–(BUSINESS WIRE)–TAI Diagnostics, Inc., focused on developing innovative diagnostic tests for monitoring the health of transplanted organs, today announced the publication, “Effect of endomyocardial biopsy on levels of donor-specific cell-free DNA” in the October, 2019 issue of The Journal of Heart and Lung Transplantation. The clinical study presented in the paper was conducted jointly […]
Abiomed Announces Q2 FY 2020 Revenue of $205 Million and 29.4% Operating Margin
DANVERS, Mass.–(BUSINESS WIRE)–Abiomed, Inc. (NASDAQ: ABMD), a leading provider of breakthrough heart recovery and support technologies, today reported second quarter fiscal 2020 revenue of $205.0 million, an increase of 13% compared to revenue of $181.8 million for the same period of fiscal 2019. Operating income was $60.2 million, up 20%, compared […]