CALGARY, Alberta, Feb. 25, 2019 (GLOBE NEWSWIRE) — Hemostemix Inc. (“Hemostemix” or the “Company”) (TSX VENTURE: HEM; OTCQB:HMTXF) is pleased to announce that it has received all approvals from UC Davis Health (“UC Davis”) located in Sacramento, California to be the clinical trial site for the Company’s Phase II clinical trial […]
Other News
REVA Announces Extension to Voluntary Suspension of Trading
SAN DIEGO, Feb. 24, 2019 (GLOBE NEWSWIRE) — REVA Medical, Inc. (ASX: RVA) (“REVA” or the “Company”) said today that on 25 February 2019 (AEDT), the Australian Securities Exchange issued a market announcement indicating that the securities of the Company will remain suspended from quotation at the Company’s request until […]
Prominent Cardiovascular Expert, James J. Ferguson, M.D., FACC, FAHA Joins Matinas BioPharma as Chief Medical Officer
BEDMINSTER, N.J., Feb. 25, 2019 (GLOBE NEWSWIRE) — Matinas BioPharma Holdings, Inc. (NYSE AMER: MTNB), a clinical-stage biopharmaceutical company, today announced that it has appointed a prominent cardiovascular medical and clinical development expert, James (“Terry”) J. Ferguson, M.D., FACC, FAHA, as its Chief Medical Officer. Dr. Ferguson is a well-recognized, industry leading […]
Quantum Genomics Completes Enrollment in Its Pharmacokinetic Clinical Study of Firibastat
PARIS and NEW YORK, Feb. 25, 2019 (GLOBE NEWSWIRE) — Quantum Genomics (Euronext Growth – FR0011648971 – ALQGC), a biopharmaceutical company specializing in the development of a new drug class that directly targets the brain to treat resistant hypertension and heart failure, today announced it has completed enrollment in its pharmacokinetic study […]
Novartis to pursue transformational therapy to reduce risk of cardiovascular disease in people living with elevated levels of inherited lipoprotein(a)
Basel, February 25, 2019 – Novartis announced today that it is exercising its option to license the rights to develop and commercialize TQJ230, an investigational agent previously known as AKCEA-APO(a)-LRx, from Akcea Therapeutics, an affiliate of Ionis Pharmaceuticals, for targeted cardiovascular therapy. TQJ230 was discovered by Ionis and has been co-developed to […]
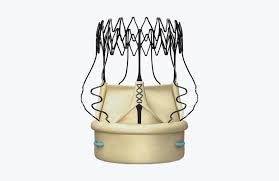
4C Medical’s Novel Mitral Regurgitation Therapy to be Highlighted at CRT 2019 Meeting
BROOKLYN PARK, Minn., Feb. 25, 2019 /PRNewswire/ — 4C Medical Technologies, Inc. (4C Medical), a developer of minimally invasive technologies for structural heart disease, today announced that its AltaValve™, a transcatheter mitral valve replacement (TMVR) technology, will be highlighted in the following presentations at the Cardiovascular Research Technologies (CRT) meeting held March 2-5, 2019 in Washington, […]
Texas Cardiac Arrhythmia Institute at St. David’s Medical Center enrolling patients to test stroke-reducing device
AUSTIN, Texas, Feb. 25, 2019 /PRNewswire/ — The Texas Cardiac Arrhythmia Institute at St. David’s Medical Center recently began enrolling patients to participate in the DIAMOND-AF II clinical trial to examine the effectiveness of the DiamondTemp System in treating patients with persistent atrial fibrillation, or A Fib. A Fib is a type of […]
Nihon Kohden Appoints New CEO of North American Operations
IRVINE, Calif.–(BUSINESS WIRE)–Nihon Kohden, a U.S. market leader in precision medical products and services, today announced the appointment of Yasuhiro Yoshitake as its new president and CEO. Yoshitake is taking over the helm of the organization from Dr. Wilson P. Constantine, who led the organization from April 2015 to February […]
LivaNova’s Perceval Sutureless Aortic Heart Valve Receives National Reimbursement in Japan to Treat Patients with Aortic Valve Disease
LONDON–(BUSINESS WIRE)–LivaNova PLC (NASDAQ:LIVN), a market-leading medical technology company, today announced that Japan’s Ministry of Health, Labour and Welfare granted national reimbursement for the Company’s Perceval® sutureless aortic heart valve to treat aortic valve disease. By adding Perceval into Japan’s health insurance system, physicians and patients have greater access to this […]
Integer Holdings Corporation Reports Results for Fourth Quarter and Full Year 2018
PLANO, Texas, Feb. 21, 2019 (GLOBE NEWSWIRE) — Integer Holdings Corporation (NYSE:ITGR), a leading medical device outsource manufacturer, today announced results for the fourth quarter and fiscal year ended December 28, 2018. Fourth Quarter 2018 Highlights GAAP Sales from continuing operations increased 0.3% and Non-GAAP Sales from continuing operations increased […]