LAVAL, Quebec, Feb. 21, 2019 (GLOBE NEWSWIRE) — Acasti Pharma Inc. (“Acasti” or the “Company”) (NASDAQ: ACST – TSX-V: ACST), a biopharmaceutical innovator focused on the research, development and commercialization of its prescription drug candidate CaPre® (omega-3 phospholipid) for the treatment of severe hypertriglyceridemia, announces it has been recognized by […]
Other News
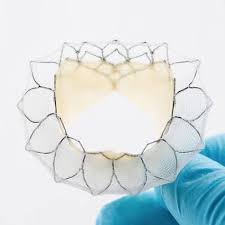
Neovasc Celebrates 5-Year Anniversary of Tiara Patient as the Longest Surviving Transcatheter Mitral Valve Replacement in the World
VANCOUVER, Feb. 21, 2019 /PRNewswire/ – Neovasc Inc. (“Neovasc” or the “Company”) (NASDAQ: NVCN) (TSX: NVCN), a leader in the development of minimally invasive transcatheter mitral valve replacement technologies and in the development of minimally invasive devices for the treatment of refractory angina, today announced that a patient implanted with a Tiara™ transcatheter […]
Rebirth of a Heart Valve: Franciscan Health Surgeon First in World to Use Device
INDIANAPOLIS, Feb. 21, 2019 /PRNewswire/ — A patient who received a first-in-the-world tricuspid valve procedure as part of a new national clinical trial at Franciscan Health Heart Center is home and recovering well. The procedure utilizes a revolutionary material called extracellular matrix. The material provides a seamless valve structure which engages and […]
Teleflex Reports Fourth Quarter and Full Year 2018 Results; Provides 2019 Guidance
WAYNE, Pa.–(BUSINESS WIRE)–Teleflex Incorporated (NYSE: TFX) (the “Company”) today announced financial results for the fourth quarter and full year ended December 31, 2018. Fourth quarter 2018 net revenues were $641.6 million, an increase of 7.8% compared to the prior year period. Excluding the impact of foreign currency exchange rate fluctuations, […]
$100 Million Invested in Clinical Research for Impella
DANVERS, Mass.–(BUSINESS WIRE)–In a milestone for scientific research, Abiomed (NASDAQ: ABMD) has now invested more than $100 million over the past five years in clinical research on the Impella heart pump platform. Abiomed’s commitment to clinical research is detailed on a new webpage that launched today. Abiomed-sponsored research is augmented by two decades of independent physician-led […]
Cardiovascular Systems, Inc. to Present at the 8th Annual SVB Leerink Global Healthcare Conference
ST. PAUL, Minn.–(BUSINESS WIRE)–On Wednesday, February 27, 2019, Cardiovascular Systems’ (NASDAQ: CSII) Chairman, President and Chief Executive Officer, Scott Ward, and Chief Operating Officer, Rhonda Robb, will present at the 8th Annual SVB Leerink Global Healthcare Conference at 3:00 p.m. Eastern Time. You may access the live audio webcast, as well […]
CHF Solutions, Inc. Announces 80% Revenue Growth for its Fourth Quarter and Provides Company Update
EDEN PRAIRIE, Minn., Feb. 20, 2019 (GLOBE NEWSWIRE) — CHF Solutions, Inc. (Nasdaq: CHFS) announced today its results for the fourth quarter and year ended December 31, 2018, which included the following highlights: Revenue for the fourth quarter ended December 31, 2018 increased 80.4 percent compared to the same period […]
BioSig Announces Successful First-In-Human Use of PURE EP System
Santa Monica, CA, Feb. 20, 2019 (GLOBE NEWSWIRE) — BioSig Technologies, Inc. (NASDAQ: BSGM), a medical device company developing a proprietary biomedical signal processing platform designed to address an unmet technology need for the electrophysiology (EP) marketplace, today announced that the Company successfully conducted first patient cases using PURE EP(tm) System, its […]
ARCA Biopharma Announces FDA Agreement for a Single Phase 3 Clinical Trial to Support Approval for the First Genetically-Targeted Cardiovascular Drug
WESTMINSTER, Colo., Feb. 20, 2019 (GLOBE NEWSWIRE) — ARCA biopharma, Inc. (Nasdaq: ABIO), a biopharmaceutical company applying a precision medicine approach to developing genetically-targeted therapies for cardiovascular diseases, today announced that it has reached agreement with the U.S. Food and Drug Administration (FDA) regarding a Special Protocol Assessment (SPA) on the design of a […]
REVA Announces Voluntary Suspension of Trading
SAN DIEGO, Feb. 19, 2019 (GLOBE NEWSWIRE) — REVA Medical, Inc. (ASX: RVA) (“REVA” or the “Company”) said today that on 20 February 2019 (AEDT), the Australian Securities Exchange issued a market announcement indicating that the securities of the Company will be suspended from quotation immediately at the Company’s request […]