DUBLIN – February 19, 2019 – Medtronic plc (NYSE: MDT) today announced financial results for its third quarter of fiscal year 2019, which ended January 25, 2019. The company reported third quarter worldwide revenue of $7.546 billion, an increase of 2.4 percent as reported or 4.4 percent on an organic basis, […]
Other News
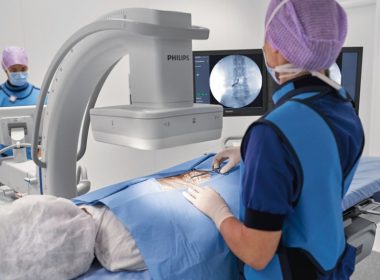
Philips launches Zenition mobile C-arm platform for enhanced operating room performance and workflow efficiency
AMSTERDAM, Feb. 18, 2019 /PRNewswire/ — Royal Philips (NYSE: PHG, AEX: PHIA), a global leader in health technology, today announced the launch of Philips Zenition, its new mobile C-arm imaging platform. Mobile C-arms are X-ray systems that are brought into the operating room (OR) to provide live image guidance during a wide range of surgeries including […]
Affluent Medical Releases Impressive Results from Scope 1 Clinical Trial Validating Efficacy of KARDIOZIS Technology
PARIS–(BUSINESS WIRE)–Affluent Medical (Paris:ALAM), a French medtech company specialized in innovative, minimally invasive implants designed to restore key physiological functions for patients suffering from heart and vascular diseases, as well as urinary incontinence, today announces positive results from its SCOPE 1 clinical trial validating the efficacy of KARDIOZIS technology. Results have been […]
Foldax, Inc. Gains Approval to Begin US Clinical Trials of the First and Only Biopolymer Heart Valve Platform
SALT LAKE CITY–(BUSINESS WIRE)–Foldax, Inc. today announced that the U.S. Food and Drug Administration (FDA) has granted IDE approval to begin an Early Feasibility Study of the Tria surgical aortic heart valve for the treatment of aortic valve disease. Using breakthrough LifePolymer™ – a proprietary advanced biopolymer material – and […]
Caladrius Biosciences Reports Top-Line Data for the Phase 2a Sanford Project: T-Rex Trial of CLBS03 for Recent Onset Type 1 Diabetes
BASKING RIDGE, N.J., Feb. 13, 2019 (GLOBE NEWSWIRE) — Caladrius Biosciences, Inc. (Nasdaq: CLBS) (“Caladrius” or the “Company”), a late-stage therapeutics development biopharmaceutical company with multiple development programs targeting select cardiovascular indications and autoimmune disease, today announced top-line results from The Sanford Project: T-Rex Study, a prospective, randomized, placebo-controlled, double-blind Phase […]
Mesoblast Financial Results Webcast for Half Year, Q2 Ended December 31, 2018
NEW YORK and MELBOURNE, Australia, Feb. 13, 2019 (GLOBE NEWSWIRE) — Mesoblast Limited (Nasdaq:MESO; ASX:MSB) will report financial results for the half year and second quarter ended December 31, 2018 in a webcast beginning at 4:30pm on Wednesday, February 20, 2019 EST; 8:30am Thursday, February 21, 2019 AEDT. The live […]
Semler to Report Fourth Quarter and Year End 2018 Financial Results and Host Conference Call on February 27, 2019
SAN JOSE, Calif., Feb. 14, 2019 /PRNewswire/ — Semler Scientific, Inc. (OTCQB: SMLR), an emerging growth company that provides technology solutions to improve the clinical effectiveness and efficiency of healthcare providers, today announced that it will report financial results for fourth quarter and full year ended December 31, 2018 before the open of U.S. […]
Medicure Provides Update on Release of Holdback Funds from Apicore Sale
WINNIPEG, Feb. 13, 2019 /PRNewswire/ – Medicure Inc. (“Medicure” or the “Company“) (TSXV:MPH,OTC: MCUJF), a cardiovascular pharmaceutical company, today announced that it has received notice from the purchaser of Medicure’s interests in Apicore of potential claims against funds held back in respect of representations and warranties under the Apicore sale agreement. The notice does […]
Boehringer Ingelheim and Lilly announce the CAROLINA® cardiovascular outcome trial of Tradjenta® met its primary endpoint of non-inferiority compared with glimepiride
RIDGEFIELD, Conn. and INDIANAPOLIS, Feb. 14, 2019 /PRNewswire/ — Boehringer Ingelheim and Eli Lilly and Company (NYSE: LLY) announced CAROLINA® (CARdiovascular Outcome study of LINAgliptin versus glimepiride in patients with type 2 diabetes) met its primary endpoint, defined as non-inferiority for Tradjenta® (linagliptin) versus glimepiride in time to first occurrence of cardiovascular death, non-fatal myocardial infarction or […]
Corindus Announces FDA Submission for Neurovascular Intervention Indication for CorPath® GRX Vascular Robotic System
WALTHAM, Mass.–(BUSINESS WIRE)–Corindus Vascular Robotics, Inc. (“Corindus” or the “Company”) (NYSE American: CVRS), a leading developer of precision vascular robotics, announced today it is seeking premarket clearance from the U.S. Food and Drug Administration (FDA) to use the CorPath GRX System in neurovascular intervention. CorPath GRX, the second generation robotic platform, received FDA clearance for percutaneous […]