BEIJING, Sept. 18, 2018 /PRNewswire/ — Huami Corporation (“Huami” or the “Company”) (NYSE: HMI), a biometric and activity data-driven company with significant expertise in smart wearable technology, today announced the launch of the new generation health band, Amazfit Health Band 1S, which delivers new features and benefits powered by AI technologies and brings advanced […]
Other News
Boston Scientific Announces Scheduled Presentations At Transcatheter Cardiovascular Therapeutics 2018
MARLBOROUGH, Mass., Sept. 17, 2018 /PRNewswire/ — Boston Scientific Corporation (NYSE :BSX ) today announced key data that will be presented at the 30th Transcatheter Cardiovascular Therapeutics (TCT), the annual scientific symposium of the Cardiovascular Research Foundation, in San Diego, on September 21 – September 25. Data from the head-to-head IMPERIAL trial evaluating the ELUVIA™ […]
DiA Imaging Analysis’ AI-Powered LVivo EF Cardiac Tool Now Available for the GE Healthcare Vscan Extend™ Handheld Mobile Ultrasound
BE’ER SHEVA, Israel, and HARTFORD, Conn., Sept. 18, 2018 /PRNewswire/ — DiA Imaging Analysis Ltd. (DiA), a provider of artificial intelligence (AI)-powered ultrasound analysis tools, announced today that its LVivo EF cardiac decision-support software is now available for purchase for GE’s new Vscan Extend™ handheld, pocket-sized ultrasound. This collaboration is an outgrowth of work between DiA […]
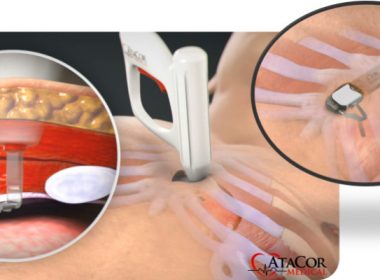
AtaCor Medical Raises $8.8M to Develop Substernal Cardiac Pacing System
SAN CLEMENTE, Calif.–(BUSINESS WIRE)–AtaCor Medical, Inc. (www.atacor.com) announced today that it has completed a $8.8M Series A financing. Co-led by Boston-based Broadview Ventures and Israel-based aMoon Ventures, the financing also includes participation from a corporate partner. The investment will support the continued development of AtaCor’s revolutionary extravascular substernal cardiac pacing […]
Contego Medical Receives 510(k) Clearance for the Paladin Carotid PTA Balloon System with Integrated Embolic Protection
RALEIGH, N.C., Sept. 18, 2018 /PRNewswire/ — Contego Medical announced today that the U.S. Food and Drug Administration (FDA) has granted 510(k) clearance for the first filter-based Integrated Embolic Protection (IEP) device, the Paladin® Carotid PTA Balloon System. Contego Medical is developing and commercializing a suite of next-generation devices that address unmet needs in […]
Medtronic Announces Clinical Study of Transcatheter Aortic Valve Replacement in Aortic Stenosis Patients with Bicuspid Valves
DUBLIN – September 18, 2018 – Medtronic plc (NYSE:MDT) today announced that the U.S. Food and Drug Administration (FDA) has approved an investigational device exemption (IDE) to initiate a single-arm study to evaluate the CoreValve Evolut(TM)TAVR system in patients with bicuspid aortic valves who are at low risk of surgical mortality. […]
LivaNova Launches International Pivotal Study Evaluating Use of Autonomic Regulation Therapy for Heart Failure
LONDON–(BUSINESS WIRE)–LivaNova PLC (NASDAQ:LIVN), a market-leading medical technology company, today announced the first successful implantation of the VITARIA® System in a patient enrolled in the Autonomic Regulation Therapy to Enhance Myocardial Function and Reduce Progression of Heart Failure With Reduced Ejection Fraction (ANTHEM-HFrEF) Pivotal Study. The study is an international, multi-center, randomized trial to evaluate the VITARIA System for the […]
Saranas™ Announces Initial Clinical Cases with the Early Bird™ Bleed Monitoring System
HOUSTON–(BUSINESS WIRE)–Saranas Inc., a medical device company with innovative technology for real-time detection and monitoring of internal bleeding during endovascular procedures, announced that the first clinical cases with Early Bird Bleed Monitoring System were performed at Atlantic Health System’s Morristown Medical Center (Morristown, NJ). The cases varied across multiple interventions […]
Freudenberg Medical Presents New Catheter Handle Platform and Steerable Introducer Products at Transcatheter Cardiovascular Therapeutics 2018
JEFFERSONVILLE, Ind.–(BUSINESS WIRE)–Freudenberg Medical, a global developer and manufacturer of finished medical devices and subassemblies, components, and minimally invasive solutions for the medical device industry, announced several new options for medical device companies looking to accelerate time to market. The new Composer®XL Deflectable Catheter Handle Platform provides a commercialization ready […]
CareDx broadens HeartCare availability
BRISBANE, Calif., Sept. 17, 2018 (GLOBE NEWSWIRE) — CareDx, Inc. (Nasdaq: CDNA), a molecular diagnostics company focused on the discovery, development, and commercialization of clinically differentiated, high-value diagnostic solutions for transplant patients, today announced the launch of the Surveillance HeartCare® Outcomes Registry (SHORE). CareDx will launch SHORE during the 22nd Annual […]