Ken Dropiewski Principal Consultant Life Science/Medical Device Practice Executive Summary: In January 2018, President Trump issued an Executive Order suspending the medical device tax (2.3% on revenue) for an additional 24 months. Further, because the moratorium is retroactive to January 1, 2018 -when the tax did commence, medical device […]
Other News
Apple Watch Series 4: Beautifully Redesigned With Breakthrough Communication, Fitness and Health Capabilities
CUPERTINO, Calif.–(BUSINESS WIRE)–Apple® today introduced Apple Watch® Series 4, redesigned and re-engineered to help users stay connected, be more active and manage their health in powerful new ways. While retaining the original iconic design, the fourth-generation Apple Watch has been refined, combining new hardware and software enhancements into a genuinely singular, […]
Infraredx to Present Results of the Lipid-Rich Plaque (LRP) Study at the Transcatheter Cardiovascular Therapeutics (TCT) 2018 Scientific Symposium
BURLINGTON, Mass.–(BUSINESS WIRE)–Infraredx, Inc., a pioneer in intravascular imaging for mapping coronary artery disease, today announced that results from the highly anticipated Lipid-Rich Plaque (LRP) Study have been accepted as a late-breaking clinical trial at the 30th Transcatheter Cardiovascular Therapeutics (TCT), the annual scientific symposium of the Cardiovascular Research Foundation, to […]
Hancock Jaffe Laboratories’ VenoValve Featured in Journal of Vascular Surgery Venous and Lymphatic Disorders as a Future Option for Treating Deep Vein Disease
IRVINE, Calif., Sept. 12, 2018 (GLOBE NEWSWIRE) — Hancock Jaffe Laboratories, Inc. (Nasdaq: HJLI, HJLIW), a company specializing in medical devices that restore cardiac and vascular health, announced today that the Journal of Vascular Surgery Venous and Lymphatic Disorders published an article on present and future options for treating deep vein disease, […]
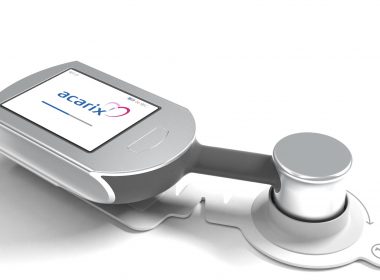
Acarix gains market acceptance in Austria; new customers orders CADScor®System for ruling out Coronary Artery Disease (CAD).
Press release Malmö, September 12, 2018 Acarix gains market acceptance in Austria; new customers orders CADScor®System for ruling out Coronary Artery Disease (CAD). Acarix AB (publ) (“Acarix”) reports first sales in Austria for its handheld CADScor®System for early rule-out of CAD. The Austrian sales mirror growing success in neighboring Germany […]
FDA to review supplemental Biologics License Application for Praluent® (alirocumab) Injection as potential treatment to reduce major adverse cardiovascular events
BRIDGEWATER, N.J. and TARRYTOWN, N.Y., Sept. 12, 2018 FDA also recently approved Praluent label update for some patients currently requiring LDL apheresis therapy /PRNewswire/ — Sanofi and Regeneron Pharmaceuticals, Inc. announced that the U.S. Food and Drug Administration (FDA) has accepted a supplemental Biologics License Application (sBLA) for Praluent® (alirocumab) Injection, a PCSK9 inhibitor. The […]
MEDTRONIC STATEMENT REGARDING RECENT FDA WARNING LETTERS
DUBLIN – September 11, 2018 – Today, Medtronic issued the following statement confirming it has received FDA warning letters (CMS #560736 and #562437) related to its Cardiac Rhythm and Heart Failure facilities located in Mounds View, Minn., and in Juncos, Puerto Rico. The warning letters reflect earlier FDA inspection reports from […]
RenalGuard Solutions Debuts Newly Formed Reprieve Cardiovascular at Heart Failure Society of America Annual Meeting and Completes $7 Million Financing
MILFORD, Mass.–(BUSINESS WIRE)–Reprieve Cardiovascular™, a newly formed company affiliated with RenalGuard Solutions™, is a pioneering medical device company focused on improving outcomes for patients suffering from acute decompensated heart failure (ADHF). RenalGuard also announced the completion of a $7 million financing provided by strong support from existing investors and Abiomed, […]
CorFlow Completes USD 9.7M in Seed Fundin
BAAR, Switzerland–(BUSINESS WIRE)–CorFlow Therapeutics AG (www.corflow-therapeutics.ch) today announced that the company has completed the 2nd close of its Seed+ financing round. To date, and since the CorFlow foundation in 2016, the company has raised a total of USD 9.7M (CHF 9.4M) in seed funding. The seed rounds have been funded by experienced private […]
With Latest FDA 510(k), physIQ Achieves Another Clearance as a Pioneer in AI Analytics
CHICAGO–(BUSINESS WIRE)–PhysIQ, a leader in applying AI analytics to biosensor data, today announced it has received FDA 510(k) clearance for its Atrial Fibrillation (AFib) detection analytics engine. The clearance extends physIQ’s portfolio of FDA 510(k) cleared analytics designed to generate clinical insight from wearable biosensors and further establishes physIQ as a […]