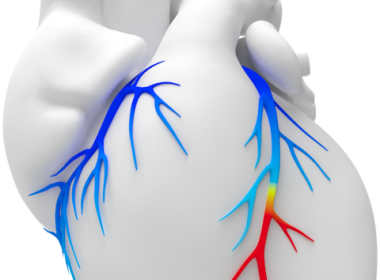
REDWOOD CITY, Calif.–(BUSINESS WIRE)–New technology using deep learning and advanced algorithms to evaluate blood flow to the heart is now being used in English hospitals to fight against coronary heart disease. Coronary heart disease (CHD) is one of the leading causes of death in the UK. It is responsible for more than […]
Other News
ROX Medical Appoints Mike MacKinnon as Chief Executive Officer
SAN CLEMENTE, Calif., Aug. 14, 2018 /PRNewswire/ — ROX Medical, Inc. (ROX), a privately held medical device company pioneering a minimally-invasive therapy for hypertension, today announced the appointment of Mike MacKinnon as its Chief Executive Officer. “Despite the many classes of drugs available to physicians to treat hypertension, millions of patients still have blood pressures […]
AngioDynamics Provides Updates on Oncology and Vascular Access Businesses
LATHAM, N.Y.–(BUSINESS WIRE)–AngioDynamics, Inc. (NASDAQ: ANGO), a leading provider of innovative, minimally invasive medical devices for vascular access, peripheral vascular disease and oncology, today provided a corporate update on enhancements made to its Oncology business and acknowledged a recent, favorable U.S. District Court ruling that impacts its Vascular Access business. […]
PaceMate™ Launches State’s First Automated Remote Cardiac Device Monitoring System at Arizona Heart Rhythm Center
BRADENTON, Fla.–(BUSINESS WIRE)–PaceMate™ is pleased to announce that as of August 15, 2018, the Arizona Heart Rhythm Center will be the first organization in the state of Arizona to offer automated remote monitoring of cardiac implantable electronic devices (CIEDs) such as pacemakers, defibrillators, and loop recorders via the PaceMate™ software […]
BioSig Technologies Announces FDA 510(k) Clearance for PURE EP System
Santa Monica, CA, Aug. 14, 2018 (GLOBE NEWSWIRE) — BioSig Technologies, Inc. (OTCQB: BSGM), announced that the Company has received 510(k) clearance for its first product, PURE EP System, from the U.S. Food and Drug Administration (FDA). The non-invasive PURE EP System is a computerized system intended for acquiring, digitizing, amplifying, filtering, measuring […]
Avinger Announces Second Quarter 2018 Financial Results
REDWOOD CITY, Calif., Aug. 13, 2018 (GLOBE NEWSWIRE) — Avinger, Inc. (Nasdaq: AVGR), a leading developer of innovative treatments for peripheral artery disease (PAD), today reported results for the second quarter ended June 30, 2018. Second Quarter and Recent Highlights Revenue of $2.1 million for the second quarter of 2018, […]
Catasys to Announce Second Quarter 2018 Results and Host Conference Call on August 14, 2018
LOS ANGELES–(BUSINESS WIRE)–Catasys, Inc. (NASDAQ:CATS) (“Catasys” or the “Company”), a leading AI and technology-enabled healthcare company, today announced that the Company will be releasing financial results for its second quarter and six months ended June 30, 2018, after the closing of the stock market on Tuesday, August 14, 2018. The Company […]
Itamar Medical Reports Second Quarter 2018 Financial Results
CAESAREA, Israel, Aug. 13, 2018 (GLOBE NEWSWIRE) — Itamar Medical Ltd. (TASE: ITMR), a company that develops, manufactures, and markets non-invasive diagnostic medical devices for sleep-apnea with a focus on the cardiology market, today reported financial results for the second quarter of 2018. “Itamar Medical continues to demonstrate double-digit growth […]
BioSig Technologies Completes Private Placement
Santa Monica, CA, Aug. 13, 2018 (GLOBE NEWSWIRE) — BioSig Technologies, Inc. (OTCQB: BSGM), a medical device company developing a proprietary biomedical signal processing platform designed to address an unmet technology need for the $4.6 billion electrophysiology (EP) marketplace, today announced that the Company closed a private placement of $4,281,000. Combined with the […]
Medicure Announces FDA Approval of Acute Care Cardiovascular Drug Sodium Nitroprusside Injection
WINNIPEG, Aug. 13, 2018 /PRNewswire/ – Medicure Inc. (“Medicure” or the “Company”) (TSXV:MPH, OTC:MCUJF), a cardiovascular pharmaceutical company, is pleased to announce that the United States Food and Drug Administration (“FDA”) has approved its Abbreviated New Drug Application (“ANDA”) for Sodium Nitroprusside Injection 50 mg/2 mL (25 mg/mL) single dose vial (“SNP”), […]