WESTMINSTER, Colo., Aug. 09, 2018 (GLOBE NEWSWIRE) — ARCA biopharma, Inc. (Nasdaq:ABIO), a biopharmaceutical company applying a precision medicine approach to developing genetically-targeted therapies for cardiovascular diseases, today reported financial results for the quarter ended June 30, 2018. “The second quarter of this year saw an important milestone for the […]
Other News
BioCardia, Inc. Reports Second Quarter 2018 Financial Results and Business Highlights
SAN CARLOS, Calif., Aug. 9, 2018 /PRNewswire/ — BioCardia®, Inc. [OTC: BCDA], a leader in the development of comprehensive solutions for cardiovascular regenerative therapies, today reported financial results and business highlights for the second quarter 2018 and filed its quarterly report on Form 10-Q for the three and six months ended June 30, 2018 with […]
NuCryo Vascular Announces Commercialization Agreement with Lokai Medical for Innovative Treatment for Peripheral Arterial Disease
SAN JOSE, Calif.–(BUSINESS WIRE)–NuCryo Vascular today announced that the company has signed a commercialization agreement with Lokai Medical, a specialty distributor of coronary/peripheral and interventional devices, to distribute the PolarCath™ Balloon Dilatation System in the United States. Balloon Cryoplasty® has been shown in clinical studies and in everyday procedures to be […]
Garmin® Health and Cardiogram announce integration of Cardiogram Heart Health app and Garmin wearables
OLATHE, Kan.–(BUSINESS WIRE)–Garmin International, Inc., a unit of Garmin Ltd. (NASDAQ: GRMN), today announced that the Cardiogram heart health app is now compatible with all Garmin wearables featuring optical heart rate, including many of the latest fēnix®, Forerunner®, and vívo series devices. The Cardiogram app provides advanced insights based on […]
Hancock Jaffe Laboratories Receives Medical Research Committee Approval for First-in-Human VenoValve Study
IRVINE, Calif., Aug. 09, 2018 (GLOBE NEWSWIRE) — Hancock Jaffe Laboratories, Inc. (Nasdaq: HJLI, HJLIW), a company specializing in bioprosthetic medical devices that establish improved standards of care for treating cardiac and vascular diseases, today announced that it has received approval for the first-in-human testing of its VenoValve® bioprosthetic medical […]
Cardiometabolic Health Congress Presents Senior Planning Committee & Chairs
Boca Raton, Florida, Aug. 09, 2018 (GLOBE NEWSWIRE) — CMHC is proud to announce its finalized Senior Planning Committee & Chairpersons for the 2018-2019 season. CMHC has cultivated a groundbreaking educational planning team, with world-renowned professors, clinicians, and researchers: all of whom will help deliver the most current, clinically accurate, and scientifically […]
Medtronic Begins Pilot Study of Investigational Extravascular ICD System
DUBLIN – August 9, 2018 – Medtronic plc (NYSE:MDT) today announced the start of a pilot study of its investigational Extravascular Implantable Cardioverter Defibrillator (EV ICD) system, in which a lead is placed outside of the heart and veins to deliver lifesaving defibrillation and antitachycardia pacing therapy all in one system, […]
Neovasc Announces Second Quarter 2018 Financial Results
VANCOUVER, Aug. 8, 2018 /PRNewswire/ – Neovasc, Inc. (“Neovasc” or the “Company”) (NASDAQ: NVCN) (TSX: NVCN), a leader in the development of minimally invasive transcatheter mitral valve replacement technologies and in the development of minimally invasive devices for the treatment of refractory angina, today reported financial results for the second quarter ended June 30, […]
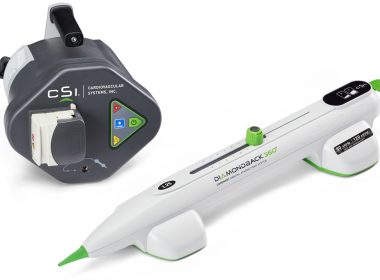
Cardiovascular Systems Presents Liberty 360° Two-Year Outcomes at 2018 Amputation Prevention Symposium
ST. PAUL, Minn.–(BUSINESS WIRE)–Cardiovascular Systems, Inc. (CSI®) (NASDAQ: CSII), a medical device company developing and commercializing innovative interventional treatment systems for patients with peripheral and coronary artery disease, presented two-year outcomes from its LIBERTY 360° study in a late-breaking presentation at the 2018 Amputation Prevention Symposium (AMP) in Chicago. The […]
CERENOVUS Receives European Regulatory Approval For BRAVO Flow Diverter to Treat Intracranial Aneurysms.
BRUSSELS–(BUSINESS WIRE)–CERENOVUS, part of the Johnson & Johnson Medical Devices Companies, announced today it has received European CE mark approval for its BRAVO Flow Diverter. The device is approved for use in the treatment of patients suffering from intracranial aneurysms. The device will divert blood flow from the aneurysm and […]