WAYNE, Pa.–(BUSINESS WIRE)– Teleflex Incorporated (NYSE: TFX), a leading global provider of medical technologies for critical care and surgery, today announced that more than 200,000 ClosureFast® Catheters have been successfully reprocessed by the company’s partner, Northeast Scientific, Inc. (NES), since NES received FDA clearance for reprocessing the popular vein ablation catheter in 2011. […]
Other News
Centerline Biomedical Completes Fifth Preclinical Study
CLEVELAND, Jan. 4, 2018 /PRNewswire/ — As Centerline Biomedical, Inc. prepares to meet with investors, strategic partners and companies in the healthcare industry during the JP Morgan Healthcare Conference, it successfully completed a fifth preclinical study at Cleveland Clinic facilities evaluating its surgical navigation system, IOPS, the Intra-Operative Positioning System. This study […]
Neovasc Reports Tiara’s Clinical Case Load Accelerating
VANCOUVER, Jan. 8, 2018 /PRNewswire/ – Neovasc Inc. (“Neovasc” or the “Company“) (NASDAQ, TSX: NVCN) today provided an update on its clinical program for the Tiara™ (“Tiara”) valve, a self-expanding mitral bioprosthesis for transcatheter implantation in patients with Mitral Regurgitation (“MR”), one of the most prevalent valvular heart diseases in western countries. MR […]
Endotronix Announces Successful First-in-Human Implantation of the Cordella Pulmonary Artery Pressure Sensor and Initiation of the SIRONA Clinical Trial
LISLE, Ill., Jan. 8, 2018 /PRNewswire/ — Endotronix, Inc., a medical technology company providing device and digital health solutions for heart failure management, today announced successful first-in-human implantation of the Cordella™ Pulmonary Artery (PA) Pressure Sensor and initiation of the SIRONA First-in-Human (FIH) clinical trial. Cardiologists Prof. Dr. Wilfried Mullens of Ziekenhuis Oost-Limburg and University Hasselt and […]
Advanced Bifurcation Systems Announces Scott King Joins as Chief of Compliance Ahead of FDA Application
Published: Jan 05, 2018 Los Angeles –January 4, 2017 – Advanced Bifurcation Systems (ABS) Scott King has joined the leadership team as Vice President of Regulatory Affairs. ABS is a clinical stage, medical device company whose stenting platform overcomes the limitations of current approaches for the treatment of bifurcation lesions in coronary angioplasties […]
The interventional cardiology devices market is expected to reach USD 20.85 billion by 2022 from USD 14.52 billion in 2017, at a CAGR of 7.5%
NEW YORK, Jan. 5, 2018 /PRNewswire/ — Read the full report: https://www.reportlinker.com/p04534881 The interventional cardiology devices market is expected to reach USD 20.85 billion by 2022 from USD 14.52 billion in 2017, at a CAGR of 7.5%. Factors driving the growth of this market include rising geriatric population, improving healthcare infrastructure, and increasing incidence of CVD. On the […]
Sensifree Reports Statistically Significant Concordance of Arterial Pressure Signal Waveform Between its Continuous Non-invasive Blood Pressure Monitoring Technology and Invasive Arterial Line
CUPERTINO, Calif.–(BUSINESS WIRE)– Sensifree, a medical device company developing advanced RF-based hemodynamic monitoring solutions, announces results of a clinical study showing excellent concordance between the signal morphology of its RF based sensor and invasive arterial line, the standard of care for continuous blood pressure monitoring. Clinical data from the study further […]
ABS Sets Their Sights on $20M in New Funding Led by New CFO
Published: Jan 05, 2018 Los Angeles, California, November 26, 2017 – Advanced Bifurcation Systems announced that is it seeking $20 million in new funding from outside investors. The new goal was announced in conjunction with the appointment of new CFO Ken Richards, who brings with him a strong background in both debt […]
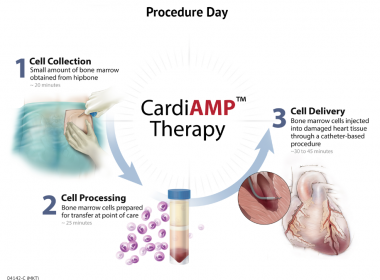
BioCardia Submits Clinical Trial to FDA for New Indication of Chronic Myocardial Ischemia for CardiAMP Cell Therapy
SAN CARLOS, Calif.–(BUSINESS WIRE)-–BioCardia, Inc. [OTC: BCDA], a leader in the development of comprehensive solutions for cardiovascular regenerative therapies, today reported filing a second Investigational Device Exemption with the FDA for the CardiAMP Chronic Myocardial Ischemia Trial to treat patients with refractory angina. This second potential indication for CardiAMP investigational cell therapy of […]
ReCor Medical Announces Completion of RADIANCE-HTN SOLO Hypertension Study
PALO ALTO, Calif., Jan. 5, 2018 /PRNewswire-USNewswire/ — ReCor Medical announced today randomization of its 146th, and final, subject in the SOLO cohort of its RADIANCE-HTN study. RADIANCE-HTN, ReCor’s study of its Paradise Renal Denervation System in people with hypertension, involves two study cohorts: “SOLO” – whose subjects are removed from anti-hypertensive […]