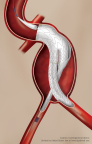
FLAGSTAFF, Ariz.–(BUSINESS WIRE)– W. L. Gore & Associates, Inc.(Gore) today announced the first implant of the GORE® EXCLUDER® Conformable AAA Endoprosthesis in the United States. The successful procedure took place on December 19, 2017 at Maimonides Medical Center in New York by Robert Rhee, MD, Chief of Vascular and Endovascular Surgery, and National […]
Other News
Avinger Announces Treatment of First Patients Globally With Next Generation Pantheris
REDWOOD CITY, Calif., Jan. 03, 2018 (GLOBE NEWSWIRE) — Avinger(NASDAQ:AVGR), a leading developer of innovative treatments for peripheral artery disease (PAD), announced that Arne Schwindt, M.D., a vascular surgeon at St. Franziskus Hospital in Münster, Germany, has successfully treated the first seven patients with the next generation Pantheris Lumivascular atherectomy system. […]
BioSig Technologies Appoints New CFO to Facilitate Growth Trajectory
Santa Monica, CA, Jan. 03, 2018 (GLOBE NEWSWIRE) — BioSig Technologies(OTCQB: BSGM), a medical device company developing a proprietary biomedical signal processing platform designed to address an unmet technology need for the $4.6 billion electrophysiology (EP) marketplace, today announced that Mr. Steven (Steve) Chaussy will be joining the company to serve in […]
Tryton Medical Names Former Johnson & Johnson Exec as New CEO
DURHAM, N.C.–(BUSINESS WIRE)– Tryton Medical, Inc., the leading developer of stents designed to definitively treat bifurcation lesions, today announced that the company has appointed Carl J. St. Bernard as president and chief executive officer. St. Bernard has a long track record growing new and established businesses in pharmaceuticals, consumer health products, […]
Investors Irate as J.P. Morgan Bars Press From Biggest Biotech Conference of the Year
The J.P. Morgan Healthcare Conference is held every year in early January, and this year it will be Jan. 8-11 in San Francisco, as usual. What isn’t quite as usual, and has some journalists and investors concerned, is a change in policy that limits media coverage. The meeting is a major event in […]
BIOLIFE4D Names Dr. Ravi Birla as Its Chief Science Officer
CHICAGO, Jan. 02, 2018 (GLOBE NEWSWIRE) — BIOLIFE4D, a biotech pioneer leveraging advances in tissue engineering to 3D print human organs viable for transplant, today announced it has named Dr. Ravi Birla as its Chief Science Officer. Birla previously served as the Associate Director of the Department of Stem Cell Engineering […]
RenalGuard System Demonstrates Promising Results from First-in-Man Studies to Evaluate Technology for Heart Failure Patients
MILFORD, Mass.–(BUSINESS WIRE)– RenalGuard Solutions, Inc., a medical device company focused on innovative technologies for the cardiac and vascular markets, today reported positive results from a first-in-man feasibility study focusing on a novel use of the RenalGuard System® to manage fluids during diuretic therapy in congestive heart failure patients suffering from fluid […]
PAVmed Announces Temporary Offer Allowing Warrants to Be Exercised at a Reduced Price
NEW YORK–(BUSINESS WIRE)– PAVmed (Nasdaq: PAVM, PAVMW) (the “Company”), a highly differentiated, multiproduct medical device company, today announced that it intends to temporarily reduce the exercise price of the Company’s outstanding Series W Warrants (Nasdaq: PAVMW, the “Warrants”) to $2.50 per share. The Company intends to file tender offer materials (the “Offer”) […]
BD Statement On FTC Clearance Of Bard Acquisition
FRANKLIN LAKES, N.J., Dec. 22, 2017 /PRNewswire/ — BD (Becton, Dickinson and Company) (NYSE: BDX), a leading global medical technology company, issued the following statement regarding today’s clearance by the U.S. Federal Trade Commission (FTC) for BD to acquire C. R. Bard (NYSE: BCR), contingent on the divestitures of BD’s soft tissue core […]
First Successful Clinical Implantation of Transcatheter Pulmonary Valve VenusP-Valve was Successfully Completed in Brazil
SAO PAULO, Dec. 22, 2017 /PRNewswire/ — On December 19, 2017, local time, 4 clinical implantations of transcatheter pulmonary valve VenusP-Valve were successfully completed in Brazil by Professor Carlos Pedra from Dante Pazzanese Institute of Cardiovascular Technology in Sao Paulo, Brazil. After Chile and Argentina, Brazil becomes the third Latin American country to apply the valve of Venus Medtech (Hangzhou) Inc. It also marks […]