DUBLIN – November 20, 2017 – Medtronic (NYSE: MDT) today announced U.S. Food and Drug Administration (FDA) approval and U.S. commercial launch for its portfolio of Azure(TM) pacemakers with BlueSync(TM)technology. Available in both single chamber and dual chamber models, the Azure XT MRI and Azure S MRI pacemakers offer improved longevity, estimated at 13.7 […]
Other News
LivaNova Enters into Binding Letter of Intent to Sell its Cardiac Rhythm Management Business Franchise to MicroPort Scientific Corporation for $190 Million
LONDON & SHANGHAI–(BUSINESS WIRE)–LivaNova PLC (NASDAQ:LIVN) (“LivaNova”) and MicroPort Scientific Corporation (HK:00853) (“MicroPort”) today announced that the companies have entered into a binding Letter of Intent (“LOI”) for the sale of LivaNova’s Cardiac Rhythm Management (“CRM”) Business Franchise to MicroPort for $190 million in cash. The CRM Business Franchise develops, […]
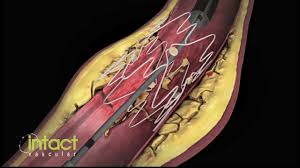
Positive Twenty-Four Month Tack Optimized Balloon Angioplasty (“TOBA”) Single Center Results Presented at VEITHsymposium™ Conference
WAYNE, Pa.–(BUSINESS WIRE)–Intact Vascular, Inc., a developer of medical devices for minimally-invasive peripheral vascular procedures, today announced that positive single center twenty-four month results from its Tack Optimized Balloon Angioplasty (TOBA) clinical study were presented at the VEITHsymposium™ 2017 conference by Dr. Christian Wissgott, Assistant Director, at Westküstenklinikum Heide in Heide, Germany. The TOBA study enrolled […]
Neovasc Closes Offering of Units Consisting of Common Shares and Warrants and Concurrent Private Placement of Notes and Warrants
VANCOUVER, Nov. 17, 2017 /PRNewswire/ – Neovasc Inc. (NASDAQ, TSX: NVCN) is pleased to announce the closing of its previously announced underwritten offering of 6,609,588 Series A units (the “Series A Units”) and 19,066,780 Series B units (the “Series B Units” and together with the Series A Units, the “Units”) of the […]
EndoVantage Secures 510(k) Clearance from FDA for SurgicalPreviewTM
SCOTTSDALE, AZ – November 8, 2017 – EndoVantage, LLC, a pioneer in cloud-based medical simulation technology, today announced its receipt of 510(k) clearance from the U.S. Food and Drug Administration for SurgicalPreviewTM, its preoperative planning system for the endovascular treatment of cerebral aneurysms. A cerebral aneurysm is a weak spot […]
MRI Interventions’ ClearPoint® NeuroNavigation System Achieves 2000th Procedure Milestone
IRVINE, Calif., Nov. 16, 2017 (GLOBE NEWSWIRE) — MRI Interventions, Inc. (OTCQB:MRIC) today announced the 2000th procedure completed with the ClearPoint NeuroNavigation System. The procedure was done by Dr. William Anderson at Johns Hopkins University. In the fall of 1998, a group of researchers at Johns Hopkins University imagined a novel […]
Merit Medical Coughs Up $100M for Bard, BD’s Biopsy Assets
Shares of Merit Medical Systems have jumped more than 9 percent this morning after the company announced it plunked down $100 million for two product lines belonging to Becton, Dickinson and Company (BD). BD is selling the assets in connection with its acquisition of C.R. Bard Inc.(Bard). Merit will acquire soft tissue core needle biopsy products currently […]
Neovasc Announces Results for the Third Quarter of 2017
VANCOUVER, Nov. 14, 2017 /PRNewswire/ – Neovasc Inc. (NASDAQ, TSX: NVCN) today announced financial results for the third quarter ended September 30, 2017 (all figures in U.S. dollars unless otherwise indicated). “From a product perspective, our two lead devices continue to perform well, quarter after quarter; patient after patient,” commented Neovasc CEO, […]
Aziyo Presents Positive Initial Data From Clinical Study of Biologic Extracellular Matrix Scaffold for Femoral Arterial Reconstruction
SILVER SPRING, Md.–(BUSINESS WIRE)– Aziyo Biologics, a fully integrated regenerative medicine company, today announced positive results from PERFORM, a prospective, post-market study conducted in patients undergoing femoral arterial reconstruction using the company’s biologic extracellular matrix (ECM) scaffold. There were no device-related adverse events or infections and primary patency was maintained in 97.6% of […]
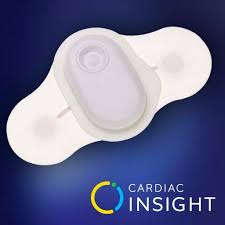
Cardiac Insight Secures First Cardiology Customers for Its Wearable ECG Sensor, Cardea SOLO™
KIRKLAND, Wash.–(BUSINESS WIRE)– Cardiac Insight, a U.S. developer of wearable medical devices and diagnostic software, announced today that the company has secured its first cardiology customers for its wearable ECG sensor device, Cardea SOLO, in Texas and several other states. Early physician and patient experiences with Cardea SOLO are confirming the […]