REDWOOD CITY, Calif.–(BUSINESS WIRE)– SentreHEART, Inc., the manufacturer of the LARIAT® Suture Delivery Device (LARIAT) will be participating in the American Heart Association (AHA) Scientific Sessions 2017 in Anaheim, California November 11-15, 2017. AHA’s Scientific Sessions attracts nearly 18,000 attendees, with a global presence from more than 100 countries. With a commitment to […]
Other News
Vascular Therapies Announces Preliminary Results From its Phase III AV Fistula Study
CRESSKILL, N.J., Nov. 9, 2017 /PRNewswire/ — An innovative, investigational treatment approach is showing the potential to address an important, unresolved problem encountered by patients with kidney failure on dialysis. At the recent annual meeting of the American Society of Nephrology (ASN), Vascular Therapies, Inc. presented preliminary results relating to […]
Postmarket Surveillance Study Confirms Benefits of Cook Medical’s Zilver® PTX® Drug Eluting Stent at 12 Months in Japanese Patients
LAS VEGAS–(BUSINESS WIRE)–One-year post-market surveillance (PMS) study data from Japanese patients who received the Zilver® PTX® drug-eluting stent confirmed the benefits of the device at 12 months, researchers announced today. Japanese researchers presented the PMS study results at themeeting in Las Vegas. This was the first time the study has been presented […]
FDA Approves Stryker’s Neuroform Atlas Stent System to Treat Wide Neck Aneurysms
KALAMAZOO, Michigan, USA, Nov. 9, 2017 /PRNewswire/ — Stryker Corporation announced today that the U.S. Food and Drug Administration has approved the Neuroform Atlas™ Stent System for marketing under a humanitarian device exemption (HDE). The device is to be utilized in conjunction with neurovascular embolic coils for the treatment of wide neck, […]
Penumbra, Inc. Reports Third Quarter 2017 Financial Results
ALAMEDA, Calif., Nov. 7, 2017 /PRNewswire/ — Penumbra, Inc.(NYSE: PEN), a global healthcare company focused on interventional therapies, today reported financial results for the third quarter ended September 30, 2017. Revenue of $83.9 million in the third quarter of 2017, an increase of 24.9%, or 24.1% in constant currency1, over the third quarter of 2016. […]
Symic Bio Announces Results from the Phase 1/2 SHIELD Trial of SB-030 in Peripheral Vascular Disease
SAN FRANCISCO, Nov. 1, 2017 /PRNewswire/ — Symic Bio, a biopharmaceutical company developing novel matrix-targeting biotherapeutics, today announced results from the Phase 1/2 SHIELD trial evaluating SB-030, a locally administered therapeutic, in patients with peripheral vascular disease undergoing angioplasty. In this first-in-human, prospective, randomized, single-blind controlled study of 67 patients, SB-030 demonstrated a […]
REVA Medical Reports Third Quarter 2017 Financial Results
SAN DIEGO, Nov. 07, 2017 (GLOBE NEWSWIRE) — REVA Medical, Inc. (ASX:RVA) (“REVA” or the “Company”), a leader in bioresorbable polymer technologies for vascular medical applications, today reported financial results for the quarter ended September 30, 2017. Third Quarter 2017 Operating Results The Company initiated commercial sales of the Fantom bioresorbable scaffold […]
Endologix Reports Third Quarter 2017 Financial Results
IRVINE, Calif.–(BUSINESS WIRE)–Endologix, Inc. (NASDAQ:ELGX), a developer and marketer of innovative treatments for aortic disorders, today announced financial results for the third quarter ended September 30, 2017. “Third quarter results were in line with our expectations, despite weather related disruptions,” commented John McDermott, Endologix’s Chief Executive Officer. “We are very […]
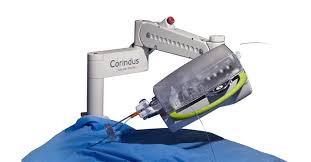
Corindus Vascular Robotics Reports Third Quarter 2017 Results
WALTHAM, Mass.–(BUSINESS WIRE)–Corindus Vascular Robotics, Inc. (NYSE American: CVRS), a leading developer of precision vascular robotics, today reported financial results for the quarter ended September 30, 2017. Recent Highlights and Accomplishments Revenue of $2.4 million in the third quarter 2017 CorPath® GRX installed base increased to 23 systems with the installation […]
Surmodics Reports Fourth Quarter Fiscal 2017 Results and Provides Fiscal 2018 Guidance
EDEN PRAIRIE, Minn.–(BUSINESS WIRE)–Surmodics, Inc. (Nasdaq: SRDX), a leading provider of medical device and in vitro diagnostic technologies to the healthcare industry, today announced results for its fiscal 2017 fourth quarter, ended September 30, 2017 and financial outlook for fiscal 2018. Recent Highlights First Patient Enrolled in TRANSCEND Pivotal Clinical […]