ATLANTA, Oct. 10, 2017 /PRNewswire/ — CryoLife, Inc. (“CryoLife”; NYSE: CRY), a leading medical device and tissue processing company focused on cardiac and vascular surgery, announced today that it has entered into a definitive agreement to acquire JOTEC AG (“JOTEC”). JOTEC is a German-based, privately-held developer of technologically differentiated endovascular stent grafts, […]
Other News
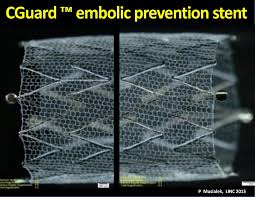
InspireMD Announces The Start Of Patient Enrollment In An Investigator Initiated Trial Of Cguard EPS In Russia
TEL AVIV, ISRAEL–(Marketwired – October 09, 2017) – InspireMD, Inc. (NYSE American: NSPR) (NYSE MKT: NSPR), a leader in Embolic Prevention Systems (EPS) / thrombus management technologies and neurovascular devices, today announced the start of patient enrollment in an investigator initiated trial in Russia, entitled: Independent Randomized Trial in Carotid Artery Revascularization Comparing […]
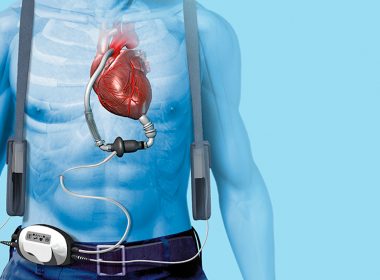
The Ethical Quandary of LVAD Disconnection
By Ken Dropiewski, Prime-Core Executive Search (ken@prime-core.com) New research brings to light a question of ethics when it comes to the deactivation of a left ventricular assist device (LVAD). Concerns differ significantly between cardiologists, hospice, and palliative medicine clinicians. This has lead to inconsistent and confusing end-of-life care for some […]
Abiomed Second Quarter Fiscal 2018 Earnings and Conference Call Notification
DANVERS, Mass., Oct. 10, 2017 (GLOBE NEWSWIRE) — Abiomed, Inc. (NASDAQ:ABMD) announced that on Thursday, October 26, 2017, the Company will release financial results for the second quarter of fiscal 2018. The Company will host a conference call to discuss the results on Thursday, October 26, 2017, at 8:00 a.m. EDT. […]
HeartStitch® GmbH Announces NobleStitch™ EL Closure Data Compared to Published PFO Clinical Trials “RESPECT” and “CLOSURE” in New England Journal of Medicine
FRANKFURT, Germany, Oct. 6, 2017 /PRNewswire/ — Prof. Dr. Achille Gaspardone, Director of Cardiology at Hospital of Sant’Eugenio (Rome, Italy) presented a comprehensive report on closure of PFO (Patent Foramen Ovale) utilizing the NobleStitch™ EL suture based closure system at the CSI-UCSF meeting on Congenital Structural Interventions in San Francisco. The NobleStitch™ EL, […]
BIOTRONIK Announces First Enrollments to BIOVITESSE Trial
(PresseBox) – BIOTRONIK has announced the start of enrollment of a coronary stent trial aiming at assessing the safety and clinical performance of a new coronary stent in de novo coronary artery lesions. On September 28, first Dr. Marco Moccetti, Cardiocentro Ticino, Lugano, Switzerland, and later on the same day Dr. Lorenz Raeber, University […]
Bay Area’s Shockwave Medical Lands $35 Million
October 10, 2017 07:00 AM Eastern Daylight Time FREMONT, Calif.–(BUSINESS WIRE)–Shockwave Medical, a pioneer in the treatment of calcified cardiovascular disease, today reported $35 million in new financing, an extension of the company’s previously announced $45 million Series C financing. New investor Fidelity Management & Research Company participated, along with […]
First Patient Treated With Microscopic Beads Pre-loaded With a Targeted Cancer Drug and Visible on CT Scans
LONDON, October 9, 2017 /PRNewswire/ — Researchers from the UCL Cancer Institute and the specialist healthcare company BTG plc (LSE: BTG) have begun the first clinical trial of an experimental treatment for liver cancer using X-ray imageable microscopic beads loaded with a targeted anti-cancer drug placed directly in the liver. The trial […]
Essential Medical Release: The MANTA Large Bore Vascular Closure Device To Be Evaluated In A 500-Patient European Post Market Clinical Registry
EXTON, Pennsylvania, Oct. 9, 2017 /PRNewswire/ –Essential Medical, Inc. today announced initiation of enrollment in a post market clinical registry in the regions where MANTA is commercially available. Principle Investigator Nicolas Van Mieghem, MD, PhD, Medical Director of the Department of Interventional Cardiology at Thoraxcenter, Erasmus Medical Center, Rotterdam, Netherlands stated, “MANTA has quickly become […]
MRI Interventions (MRIC) Names Former Royal Philips Electronics N.V. (PHG) Exec as New CEO
IRVINE, Calif., Oct. 09, 2017 (GLOBE NEWSWIRE) — MRI Interventions, Inc. (OTCQB:MRIC) today announced the appointment of Joseph Burnett as President and CEO, effective November 7. The company’s current President and CEO, Frank Grillo, will continue in the role until November 6, and will serve as an executive advisor to […]