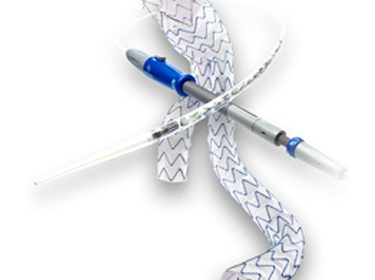
DUBLIN – October 9, 2017 – Medtronic plc (NYSE: MDT) today announced that it has received U.S. Food and Drug Administration (FDA) approval for the Endurant(TM) II/IIs stent graft system to treat abdominal aortic aneurysm (AAA) patients with neck lengths down to 4mm and <=60° infra-renal angulation when used in combination […]
Other News
Endologix Receives IDE Approval For The EVAS2 Confirmatory Clinical Study To Evaluate The Nellix Endovascular Aneurysm Sealing System
IRVINE, Calif.–(BUSINESS WIRE)–Endologix, Inc. (Nasdaq:ELGX), a developer and marketer of innovative treatments for aortic disorders, announced today that it has received Investigational Device Exemption (“IDE”) approval from the United States Food and Drug Administration (“FDA”) to commence a confirmatory clinical study to evaluate the safety and effectiveness of the Nellix […]
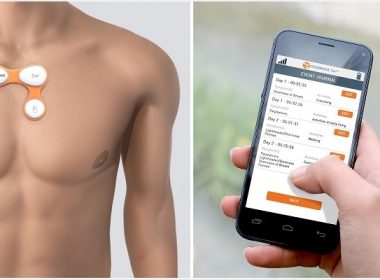
FDA Grants Marketing Clearance for the Peerbridge Cor™ Multi-channel Remote ECG Monitor
NEW YORK, Oct. 4, 2017 /PRNewswire/ — Peerbridge Health Inc., a Health IT company, announced today that its first product, the Peerbridge Cor™ System — a wireless electrocardiogram (ECG) monitor — has received 510(k) clearance from the U.S. Food and Drug Administration (FDA). The patented Peerbridge Cor has the smallest on-body footprint of any […]
Cardiologs Raises $6.4 Million to Lead the AI Revolution in Cardiology
PARIS & SAN FRANCISCO–(BUSINESS WIRE)–Cardiologs Technologies announced today that it has raised $6.4 million in a Series A financing with a syndicate of life science and technology investors (Idinvest, ISAI, Kurma Partners, Partech Ventures) and with continued support and participation from current investor Bpifrance seed fund (F3A). This round brings total […]
BTG to buy Roxwood Medical for up to $80 million
UK’s BTG to buy Roxwood Medical for up to $80 million Reuters Staff Reporting by Radhika Rukmangadhan in Bengaluru; editing by Jason Neely Our Standards:The Thomson Reuters Trust Principles. (Reuters) – British drugs company BTG Plc (BTG.L) said on Thursday it will buy U.S.-based cardiovascular catheter maker Roxwood Medical for […]
ROX Medical CONTROL HTN-2 Clinical Trial; First Procedure Performed
SAN CLEMENTE, Calif., October 3, 2017 – ROX Medical Inc. A privately held medical device company pioneering an innovative interventional vascular therapy for Uncontrolled Hypertension, announced today that the first patient was treated in the CONTROL HyperTeNsion (HTN)-2 clinical study, the Company’s pivotal study to evaluate the safety and effectiveness of […]
Bolton Medical Announces the First Implant of the Relay®Pro Clinical Trial for Treatment of Thoracic Aortic Dissections
Sunrise, Fla, October 3, 2017 – Bolton Medical announced the first patient has been enrolled in the RelayPro FDA Phase II Clinical Trial for Treatment of Acute Complicated Type B Thoracic Aortic Dissections. This trial will assess the safety and efficacy of RelayPro to treat Type B dissections. Christian Shults, M.D., Cardiothoracic […]
Cardiovascular Systems to Webcast Fiscal 2018 First-Quarter Earnings Conference Call Wednesday, October 25
ST. PAUL, Minn.–(BUSINESS WIRE)–Cardiovascular Systems, Inc. (CSI) (NASDAQ: CSII) will host a live webcast of its fiscal 2018 first-quarter conference call on Wednesday, October 25, 2017, at 3:45 p.m. CT (4:45 p.m. ET). Scott Ward, Chairman, President and Chief Executive Officer, and Larry Betterley, Chief Financial Officer, will discuss CSI’s […]
BioCardia Receives U.S. Patent Covering Morph Product Family Design
SAN CARLOS, Calif.–(BUSINESS WIRE)–BioCardia®, Inc. (OTC: BCDA), a leader in the development of comprehensive solutions for cardiovascular regenerative therapies, with clinical programs in heart failure, today announced the issuance of United States Patent No. 9,775,963 entitled, “Steerable Endoluminal Devices and Methods.” BioCardia CEO Peter Altman, PhD stated, “This new patent […]
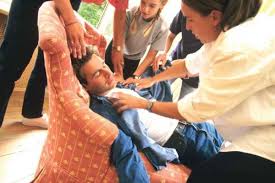
BIOTRONIK CLS Technology Shown To Reduce Fainting In Syncope Patients By Seven-Fold
Robustly Positive Results of SPAIN Study Published in Journal of American College of Cardiology BERLIN, Germany, October 4, 2017 – BIOTRONIK’s exclusive rate responsive technology, Closed Loop Stimulation (CLS) has shown to reduce fainting in pacemaker patients with vasovagal syncope up to sevenfold. Results from the SPAIN study, the first […]